More Information
Submitted: March 03, 2026 | Accepted: April 08, 2026 | Published: April 10, 2026
Citation: Kavita, Tewari S. Listeria monocytogenes: A Tale of Food-Borne Pathogen. Arch Case Rep. 2026; 10(4): 30-38. Available from:
https://dx.doi.org/10.29328/journal.acr.1001186
DOI: 10.29328/journal.acr.1001186
Copyright license: © 2026 Kavita, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Keywords: Listeria monocytogenes; Food borne pathogen; Listeriosis; Listeriolysin O; Robustness; Virulence factors; Biofilms
Abbreviations: LM: Listeria monocytogenes; FDA: Food and Drug Administration; CDC: Centers for Disease Control and Prevention; C- met: Mesen chymal–epithelial transition factor; AI: Artificial Intelligence
Listeria monocytogenes: A Tale of Food-Borne Pathogen
Kavita and Sakshi Tewari*
Department of Life Sciences, J.C. Bose University of Science and Technology, YMCA, NH-2, Sector-6, Mathura Road, Faridabad, Haryana, 121006, India
*Corresponding author: Sakshi Tewari, Department of Life Sciences, J.C. Bose University of Science and Technology, YMCA, NH-2, Sector-6, Mathura Road, Faridabad, Haryana, 121006, India, Email: [email protected]; [email protected]
Listeria monocytogenes is one of the major foods borne pathogen that is found in soil, water and animal faeces which causes the disease known as listeriosis. The pathogen mainly affects the newborns, pregnant women, elderly age people, and immunocompromised host. It mainly enters in the gut of humans through the intake of raw vegetables, unwashed foods, uncooked or half cooked food. It is difficult to recognize the bacteria as it does not bring immediate change in the food color and food taste. This review aims to highlight the mechanism of infection of the bacteria, its special characteristics, symptoms of the disease, pathogenesis and treatment prevention.
Graphical abstract
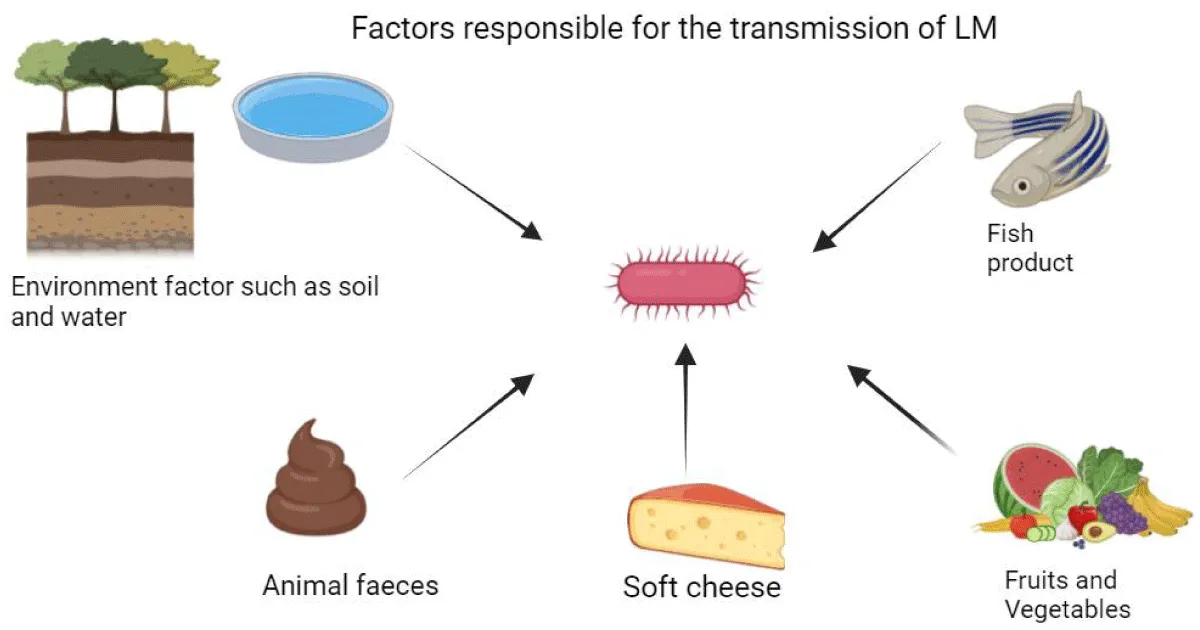
Listeria monocytogenes is a Gram-positive, rod shaped, non-spore forming bacteria that appears coccoid and motile having peritrichous flagella. It can also appear in single or short chains V form. This bacterium has ubiquitous nature, due to which it is a food safety threat and can easily enter the processing environment through raw ingredients [1]. It can be found in soil, water, vegetation, animal faeces, soft chesses, and cold smoked fishery products (Figure 1).
Figure 1: Factors contributing to L. monocytogenes transmission, and this figure was created by BioRender.
It has the capability to grow in refrigerated, packaged, ready-made products without changing food’s taste or smell. They are 0.5 - 2 micrometer in length and 0.5 - 4 micrometer in diameter. Listeria is observed positive for various biochemical tests including catalase, [2] Christie-Atkins Munch-Peterson (CAMP), β hemolysis, Methyl-Red-Voges-Proskauer (MRVP), [3]. It can survive at optimum temperature of 30 ℃ - 37 ℃, can grow beyond 45 ℃ and pH range of 5.5 - 9.6. The pathogen can tolerate high salt concentration of 10% - 12% NaCl [4] and is able to survive and replicate in somatic as well as non-somatic cell. This food-borne pathogen is heat resistant, so it can even display growth beyond these temperature ranges. Listeriosis is a very dangerous disease which affects the people by eating the contaminated food. Symptoms in the infected people appears with in few hours and can be appear in two days after eating the contaminated food. Severe form of the bacteria takes days to months to show the symptoms. Listeriosis can affects the various organs/sites of the human body including: brain, spinal cord membranes, blood etc. Invasion of Listeria in human body could be of two types non-invasive listeriosis and severe infection. Non-invasive includes moderate form of the disease including various symptoms like fever, diarrhea, headache and muscle pain. Severe infection mainly affects pregnant women and people with weakened immune system. Disease has wide range of symptoms, it mainly depends on the severity of illness, symptoms may last from days to weeks. Mild symptoms include: fever, muscle aches, nausea, vomiting, and diarrhea. Severe form includes stiff neck, confusion, loss of balance, and convulsions. In immuno-compromised host, old age patients, or individual suffering from health issues including diabetes, kidney disorders, lung diseases etc. May eventually lose their lives due to untreated listeriosis.
Outbreaks of L. monocytogenes
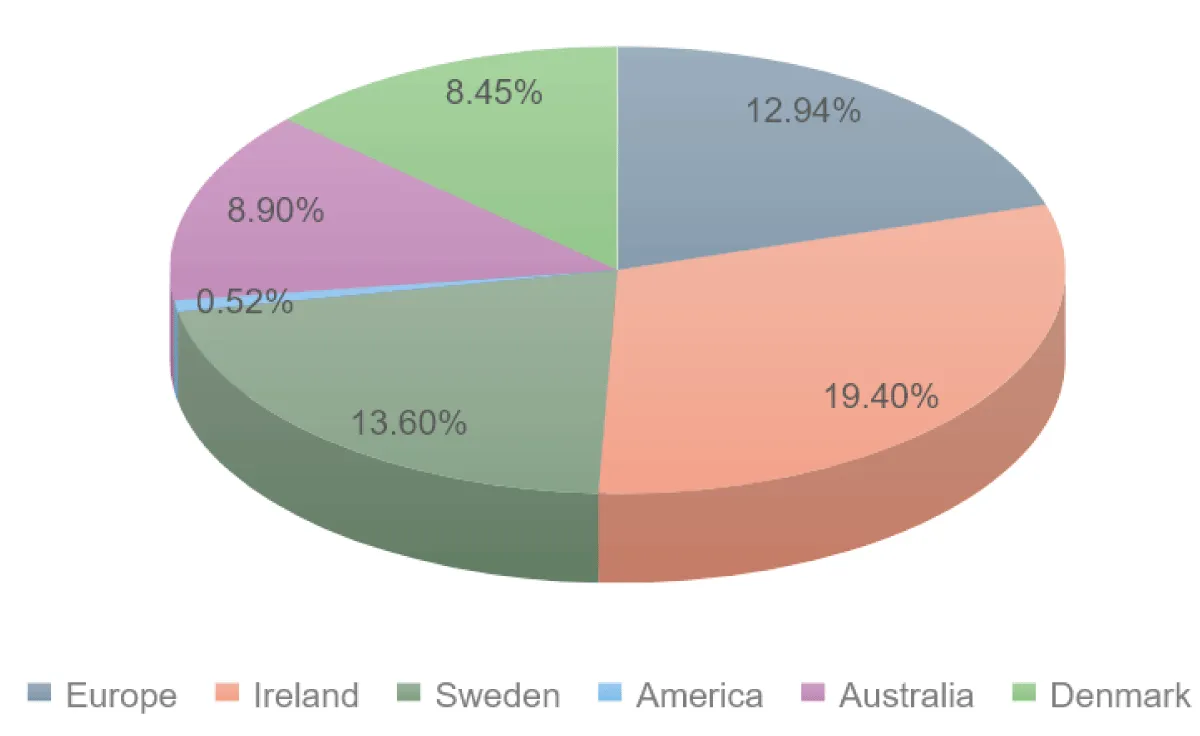
As per the international reports, Ireland reported the highest cases of listeriosis with 19.4%. The increased number of Listeria infections in Ireland has no bearing on a particular significant issue. The high percentage could be caused due to several factors, such as the type of bacteria, temperature, improved healthcare surveillance, the population of Ireland, and the increased demand for packaged food, including ready-to-eat food items. Listeria is a psychotropic bacterium that survive at 11 °C which facilitates bacterial survival in Ireland’s cold climate. Increased incidences also shows that the country has an improved healthcare surveillance system that can identify even extremely low mild cases. Over 15% of Ireland’s population is elderly, which is a significant contributing factor to bacterial infections followed by Sweden with 13.6%, Australia with 8.90%, Denmark with 8.45%, Europe with 2.94%, America with 0.52% (Figure 2) [5].
Figure 2: International outbreaks of listeriosis in various countries.
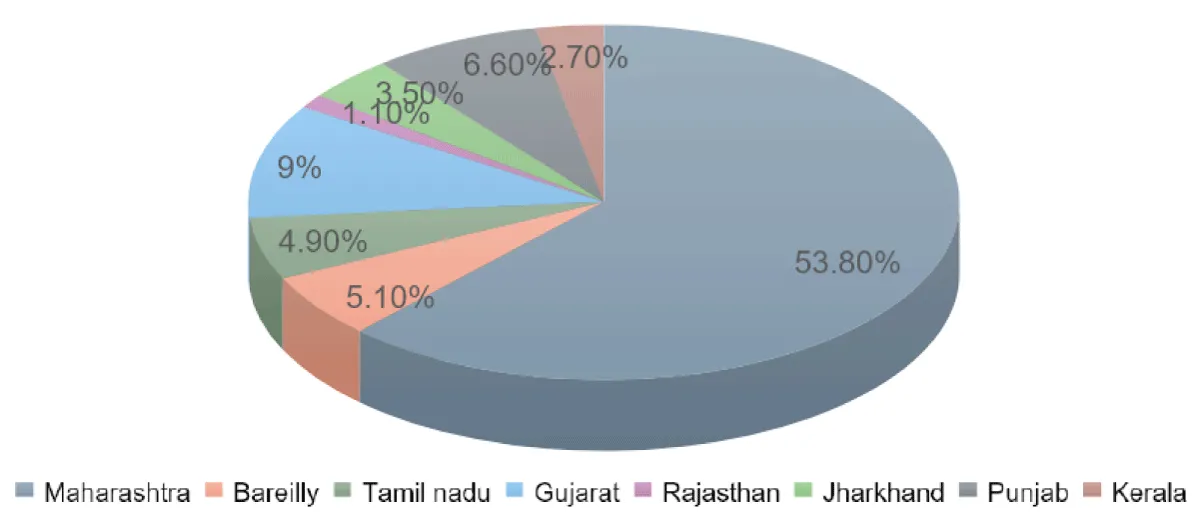
As per the global data, published in 2017, 8 states of India reported high outbreaks of L. monocytogenes. Maharashtra reported highest cases of listeriosis 53.8% followed by Gujarat with 9%, Punjab with 6.6%, Bareilly with 5.1%, Tamil Nadu with 4.9%, Jharkhand with 3.5%, Kerala with 2.1%, Rajasthan reported with the least cases of about 1.1% (Figure 3).
Figure 3: National outbreaks of listeriosis in different states of India during the years (2001-2018).
Human listeriosis is a dreadful disease that has impacted in India and other countries as well in last two decades. There has been a list of reports on the outbreak of Listeria. There have been more than 30 outbreaks of listeriosis in various parts of the globe since 1981 due to consuming various food products including raw and industrially processed foods. According to the data, in 2024, a deadly outbreak of Listeria in 11 states has been linked to dozens of dairy products [6], including popular super bowl snacks, queso fresco and Cotija cheese. In the America, Listeria is the third leading cause of death from foodborne illness killing about 260 people in a year (as per the CDC, 2024). Even with adequate antibiotic treatment, the disease has a high mortality rate of about 20% to 30%, according to American Food and Drug Administration. Pregnant women are about 10 times more likely to prone from listeriosis and the illness can be fatal to the foetus (as per the FDA, 2024). The two deaths associated with the infections were reported in California and Texas. Deli meat and hot dogs were common source of Listeria outbreaks in the 1990s, likely because of the difficulty in cleaning the resistant bacteria from every nook and cranny in deli food preparation devices. Eating foods made from raw milk is risky and consuming it can make person infected with Listeria and other harmful germs.
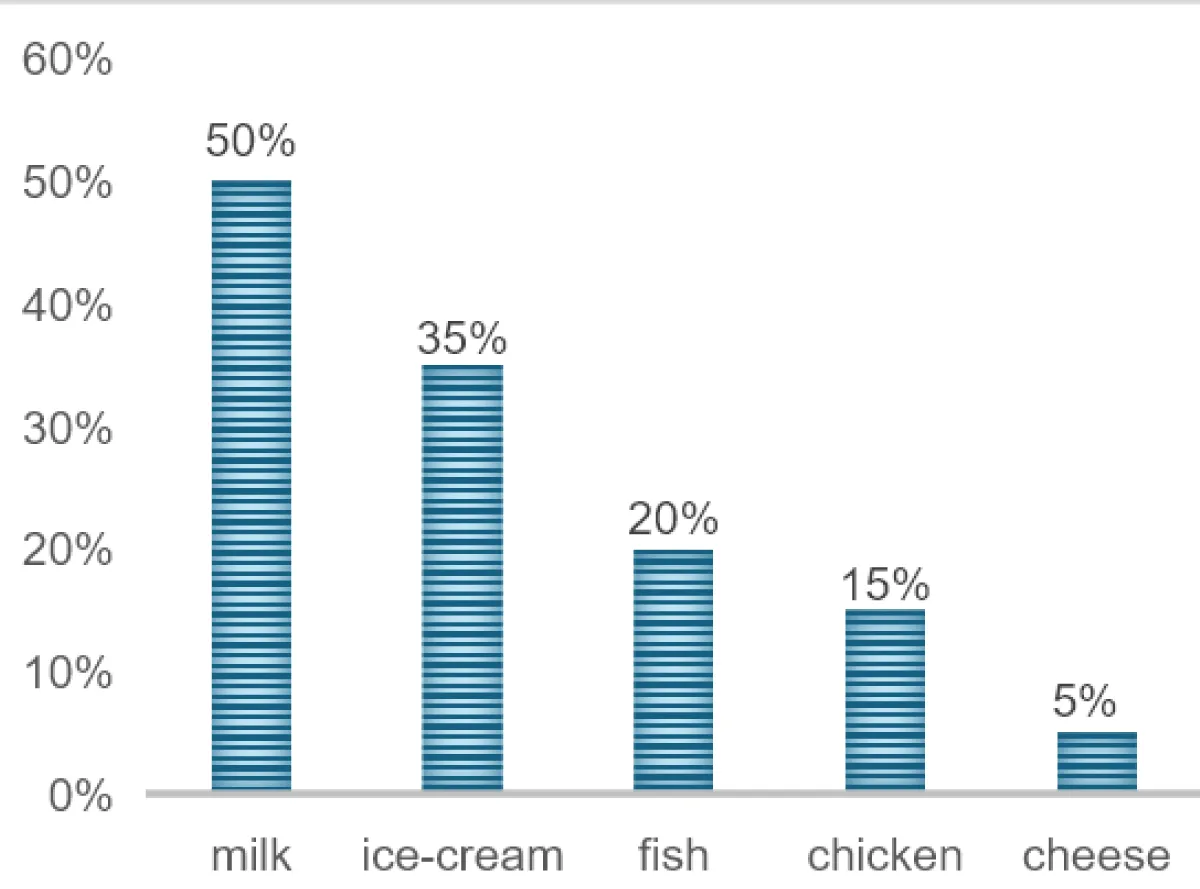
There are several food items that resulted in the outbreak of Listeria. In year 2000 raw milk [7] and raw meat served as a source for outbreak. In year 2011, 2012, 2013, 2014 Cantaloupes, Ricotta cheese, Cheese dairy product beans worked as a source of outbreak respectively (Figure 4). High-risk infection individuals should not eat raw or even lightly cooked sprouts as they may be contagious. Always thoroughly heat sprouts first - even homegrown versions can harbour the bacteria. Certain leafy greens such as kale, spinach lower to the ground and are more chances to contaminate by Listeria in the soil. Melons are another source of Listeria infection.
Figure 4: Percentage of contaminated food samples with L. monocytogenes.
Mechanism of action of L. monocytogenes
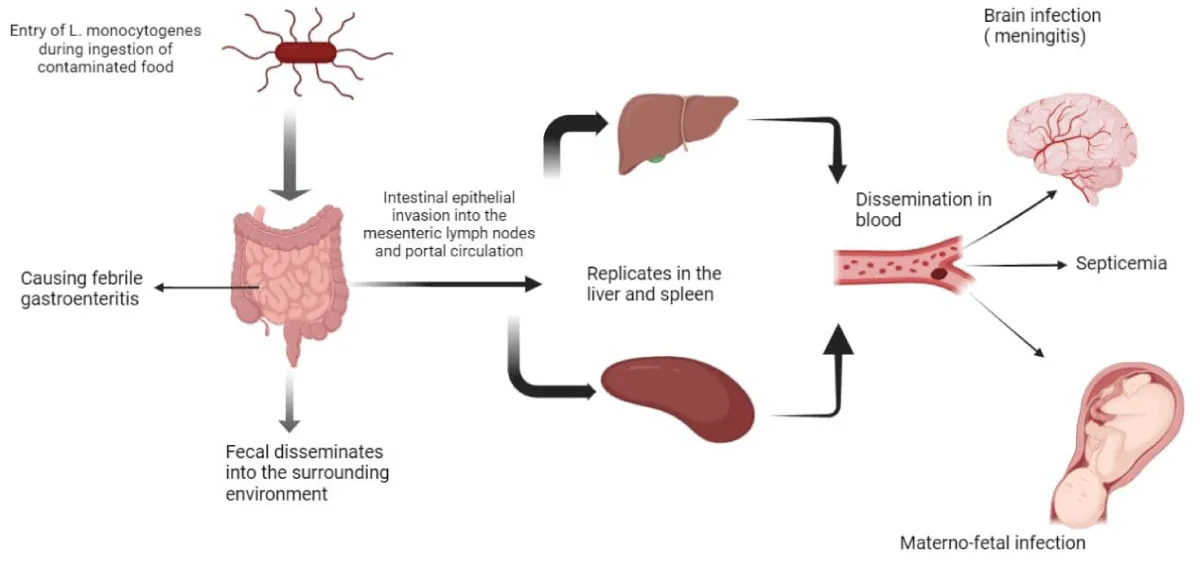
In Animal excrement, soil, and water all naturally contain Listeria. It now reaches the crop during the agricultural stages through soil and water (the bacteria enter the crops through the contaminated water). These bacteria create a biofilm inside the crops, which prevents them from being eliminated even after repeated processing. Additionally, Listeria will survive during refrigeration processing because of the temperature, which would be favorable for its growth. Finally, it enters humans through the food products which are ready to eat. There are several reports that explain the impact of L. monocytogenes on humans. Maximum findings stated that listeriosis can be specifically life-threatening for pregnant women and their infants, which may lead to serious complications with their pregnancy and can lead miscarriage and stillbirth. Infants born with listeriosis infection suffers from various complications and require immediate medication, even lead to life-long health problems and ultimately leads to death. According to the Centre for disease control (as per CDC, 2017) and prevention report 1,600 people get infected with Listeria each year in the United States and 260 people die from the infection. The bacteria could be isolated from the human digestive tract during the time of infection. Food that involves the highest risk of infection includes consumption of raw sprouts. unpasteurized milk, soft cheeses, cold deli meats, cold hot dogs, smoked seafood etc. If an individual consumes such contaminated food containing Listeria it can significantly harm the person terribly. Primary virulence factors of Listeria include Internalin (bacterial surface proteins for host cell attachments). Listeriosis O (helps the bacteria escape out from host cell vacuole). Actin-polymerization: (helps the bacteria to move within and between cells). Phosphatidylinositol-specific phospholipase C (helps the bacteria to escape from host cell vacuole and cause membrane disruption). The pathogen has the capability to grow at reduced temperatures. Low temperatures induce enzyme such as RNA helicase that modifies the bacterial activity and replication process at low temperatures. The capability of the bacteria to produce biofilms enhances the ability to survive under harsh environmental conditions. Listeria also utilizes flagella at reduction temperatures. L. monocytogenes has cell-surface galactose residues, lipoteichoic acids, and surface proteins called internalin that binds primarily to gastrointestinal epithelial cells via host protein cadherins, allowing entry into the cell. After invasion into host cells, bacteria have the ability to initiate a cell-mediated immune response in the host phagocytized, L. monocytogenes can lyse the internalized vacuole via a pore forming cytotoxic protein called Listeriolysin O among other phospholipase proteins which are not pore-forming. It is also responsible for displaying the Beta-hemolysis when grown on blood agar. Once free from the vacuole, the bacteria can disrupt the normal cellular processes by moving through the cell via actin polymerization. The bacterium surface protein, Act A [8] stimulated by host intercellular proteins that innately regulate actin filaments. These actin monomers are connected only at one end utilizing host intracellular cytoskeleton filaments. This tail, often refers as rocket tail that provide the ability of bacteria to move swiftly through the intracellular cytosol, between cells, or disseminate hematogenous. Cadherins is an epithelial attachment protein that is found in abundance in the blood-brain barrier as well as the placental-fetus barrier which may explain why the bacteria can infect neonates and cause meningitis. L. monocytogenes forms “rocket tails” via actin polymerization that allows the bacteria to move rapidly between cells, avoid antibody detection, and spread hematogenous. Once the infection has occurred L. monocytogenes can cause amnionitis, sepsis, spontaneous abortion in pregnant women, granulomatous infant septic, and meningitis.
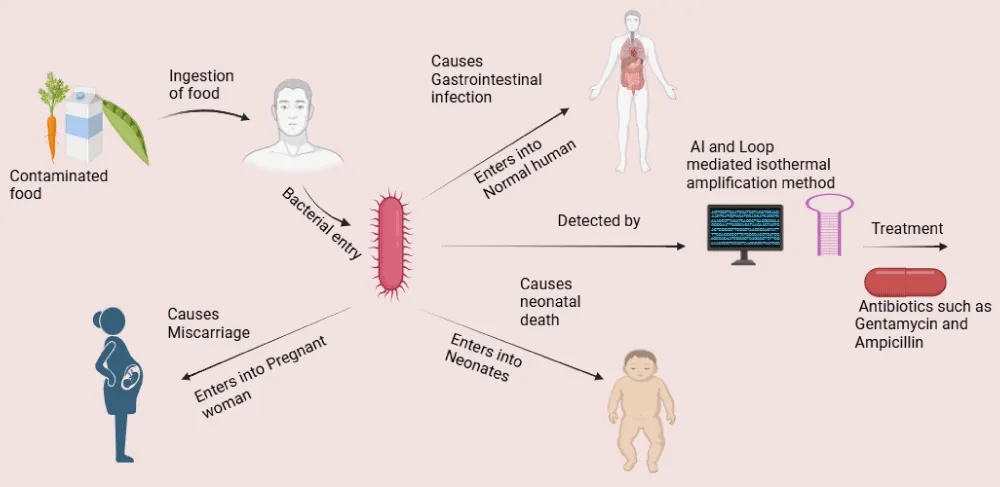
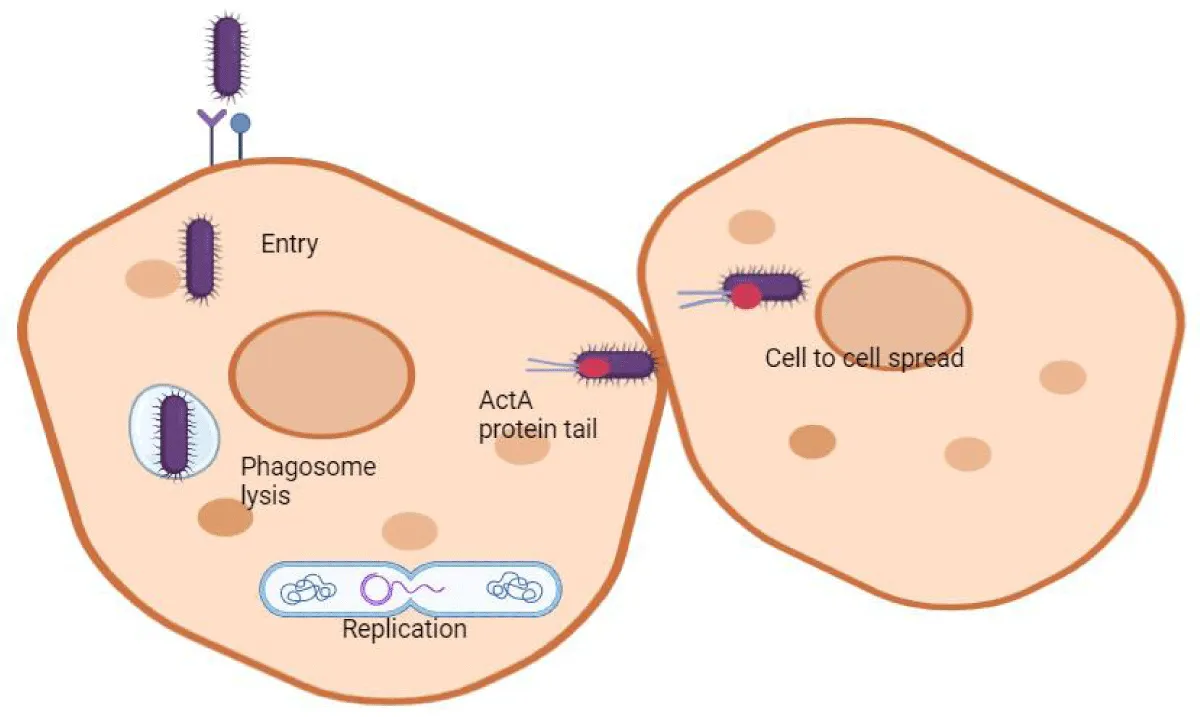
1. In normal humans: L. monocytogenes mechanism of infection involves several steps; Listeria can be transmitted into the host by the consumption of contaminated foods. Several dairy products can act as a source by which bacteria transmit in host by ingestion and bacteria primarily enter into the host intestine [8]. This is the key step when Listeria evade the host cell, two surface proteins that are used by Listeria to invade the non-phagocytic cells (liver, macrophage and some epithelial cells) includes Internalin A and B [9]. Internalin A [10] is a protein used by bacteria to bind E-cadherins on the surface of non-phagocytic cells. Internalin B [10] is used by bacteria for the interaction of C-met receptor these interactions would help the bacteria to internalized into the host cell through phagocytosis and endocytosis [11]. After entering into the host, membrane bound vesicles can easily trap the bacteria, these vesicles are called phagosome, when bacteria get trap into the phagosome [12], it releases a toxin called listeriolysin. It refers to the toxin produce by the bacteria which is used to lyse the phagosome so that phagosome is not able to release lysosome (that including enzyme lipases, nucleases, sulfatases that damage the bacteria) and prevent itself from being damaged bacteria also produces two phospholipases [12], these are phospholipase A and B which is used for the breakdown of vacuolar membrane. Once the bacteria pass the phagosome it enters into the cytoplasm bacteria could easily replicate into the cytoplasm of host cell. Bacteria expresses a protein called Act A or Actin A protein with the help of this protein a long tail of actin protein is attached at one side of the bacteria make it communicable with the neighboring cell and hence the infection spreads into the other cells. Once bacteria enter into the neighboring cell it gets trapped into the membrane bound vesicles and releases listeriolysin and the infection cycle keep goes on (Figure 5).
Figure 5: Mechanism of infection of L. monocytogenes in a normal human, and this figure was created by BioRender.
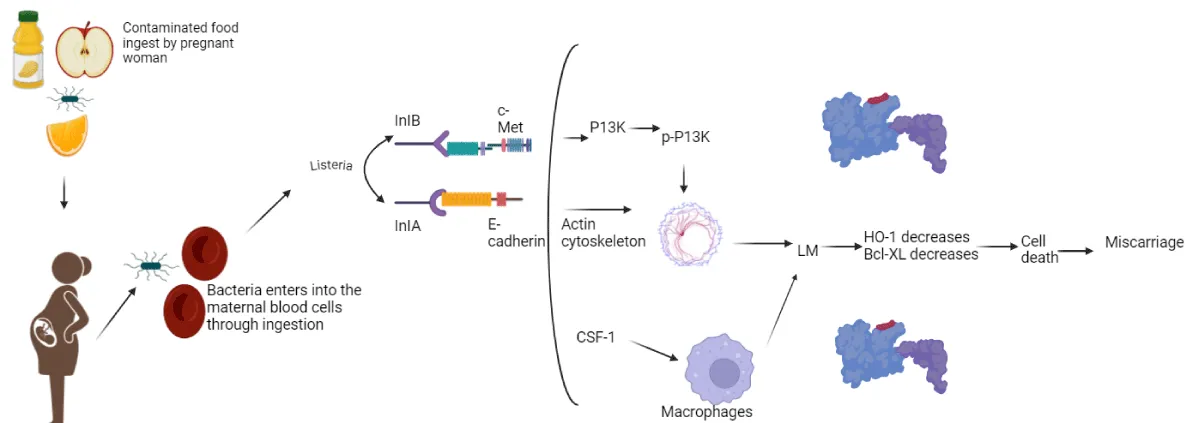
2. Pregnant women: During pregnancy, ingestion of the contaminated food, allows the bacteria enter inside the body. As the immune system is already suppressed, it does not recognize the bacteria and phagocytosis does not initiate and hence bacteria become presiding. Bacteria primarily enter into the intestine cause intestinal evasion and then migrate into blood cause bacteremia. Listeria can affect both types of cells which are extra villous trophoblast cells (EVT) [13] and syncytiotrophoblast cells (SYN) [14]. L. monocytogenes entered into the SYN of the placenta. Bacteria especially infect placenta [15] in the pregnant women to cause listeriosis because placenta is the key site to produce bacterial surface proteins Internalin A, this protein is used by the bacteria to binding of E-cadherins to the non-phagocytic cells. This binding will help the bacteria to uptake, once the uptake is done bacteria become able to cross the placental barrier. Mostly from week 29 to week 40 [16] infection become very frequently which also indicates that the SYN might be there where the bacteria enter in the placenta bacteria could instantly cohere to the trophoblast epithelium penetrate the trophoblast layer and get into the center of villous tissues [17] In the placenta bacteria primarily infects the cells which are present in the middle arterial catheter where the staining result was positive for cytokeratin stipulate its fetal trophoblastic source. Bacteria could easily migrate to other trophoblastic cells and could infect the cells of SYN villous center in the labyrinthine zone of placenta. Studies on mouse observed that pregnant mice were more prone to this bacterial infection, associated with Foxp3 and T regulatory cells [18]. Bacteria primarily infect the fetal by crossing the placental barrier, and afterwards it reduces the capability of the fetal tolerance to T regulatory cells [19]. As compared to normal female’s pregnant females are 19 times more prone to this infection because in the reduction of T cell immunity. Accumulation of maternal T cells bind with the fetal antigens and CD8+ cells increased the level of CXCR3 [20] recruiting CD8+ T cells into the decidua [21], that leads to fetal death. The bacterial infection reduced the level of B- cell lymphoma extra- large (Bcl-XL) and heme oxygenase level 1 (HO-1) [22], cells which leads miscarriage/fetal death. Besides an anti- listeria reaction depend upon colony stimulating factor (CSF –1) [23] mediated by macrophages (Figure 6).
Figure 6: Mechanism of infection of L. monocytogenes in a pregnant woman, and this figure was created by BioRender.
3. Mechanism of action in infants: Neonatal could be infected by early onset listeriosis (from placenta transmission), in pregnant women, bacteria can cross the placenta and enter into amniocentic fluid that infect the fetus. Higher mortality rate about (20% - 40%) and it is not easily diagnosed. Neonatal could be infected by late onset listeriosis (from vaginal delivery), if the bacteria are present in the vaginal region of the women during the delivery time, neonatal inhale or ingest the bacteria. Lower mortality rate about (0% - 20%) and it is easily diagnosed. When it enters into the body, it directly suppressed the functioning of immune system which reduces the T cell -immunity and body is not able to fight with the pathogen. The bacteria cause several new diseases in the infants: sepsis, granulomatosis (Figure 7).
Figure 7: Mechanism of infection of L. monocytogenes in neonates and this figure was created by BioRender.
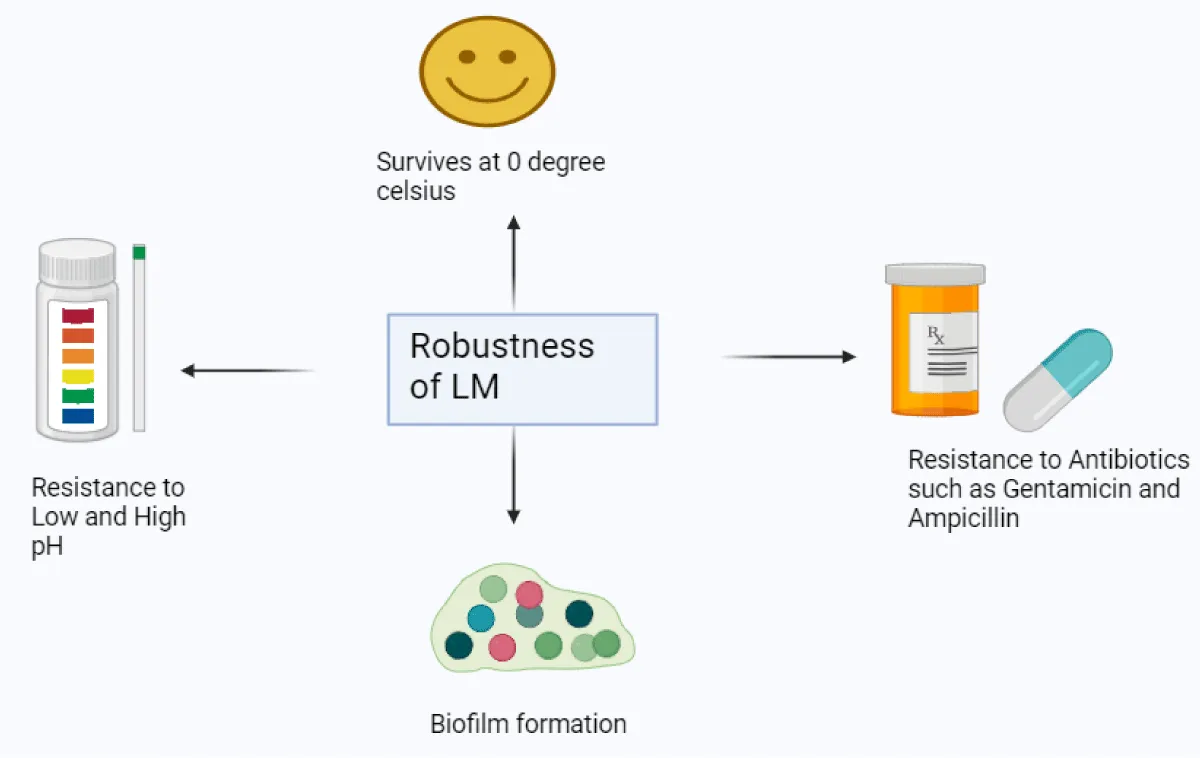
Robustness of Listeria
Listeria is quite robust and strong bacteria. Adverse conditions have little or no impact on its growth that is the major reason why has ability to cause severity. Low temperature; Listeria can survive at a temperature of 0° to 45 °C [24] at low temperature cell is affecting and as a result, lead to the reduction in the membrane fluidity, when stress conditions increases Listeria response to stress conditions by changing the membrane fluid composition towards increase concentration of unsaturated fatty acids, this shifting is important to inhibit the formation of gel like state so that no cytoplasmic leakage can occur, sigma B factor play a key role for the bacteria to adapt the environment which prefers low temperature [25] sigma B help the formation of cold shock domain family proteins,(Csps) these proteins help to inhibit the formation of misfolded proteins that generally occur in the cold stress environment so that Listeria can easily survive. Sigma B protein regulate those genes which help in the formation of compatible solutes, such as (Betaine and Carnitine). Thus, these factors help listeria for adaptation and survival at 0 ºC (Figure 8).
Figure 8: Robustness of L. monocytogenes, and this figure was created by BioRender.
Low pH: Low pH indicates that the environment is highly acidic, this high acidity refers more protons H+ ions inside a cell, so the bacteria use two defence pathways against these high H+ ions. The first defence pathway is Glutamate decarboxylase system, this is a defence mechanism where glutamate is converted into Gamma Amino Butyric Acid (GABA) with the help of the enzyme glutamate decarboxylase [26] during this conversion the H+ protons are deproteinized from the cell as deprotonation increases less H+ protons are available inside the cell, this leads to reduce the acidity inside the cell. Now listeria can easily survive inside the cell, by using the first defence pathway. The second defence pathway used by the bacteria is ADI pathway [27] where ADI stands for Arginine deaminase pathway, in this system arginine is converted into ornithine and ammonia this reaction catalyses by the enzymes carbamoyetransferase and carbamate kinase. ammonia reduces the acidity inside the cell and promote a suitable environment for the survival of Listeria.
High pH: High Ph indicates the environment is highly alkaline, high alkanity refers more OH- ions F1 – F0 ATP system, helps to remove the H+ protons inside the cell and promote the production of bufferine which is an alkaline compound, this compound maintain the alkanity inside the cell so that environment inside the cell is transient from acidic to alkaline environment for bacterial survival ,sigma B proteins also help by regulating the expression of the genes which are responsible for the maintaining of a alkaline environment.
Biofilm formation: Listeria is able to make biofilm formation through the attachment of various surfaces including; stainless steel, polystyrene and glass [28]. Listeria forms biofilms at a 4 ºC and 12 ºC. Some scientists observed that this bacteria form biofilm at a temperature of around 4 ºC with higher levels on glass instead of polystyrene and stainless steel. Some observed that formation of biofilm at higher levels on stainless steel and glass at a temperature of 4 ºC instead of polystyrene. Some concluded that formation of biofilm at higher levels on glass. Bacteria shows flagella at a temperature less than 30 ºC and become non flagellated at a temperature greater than 30 ºC. Biofilm refers cluster of bacteria attached to a surface, formation of biofilm including four steps; attachment, EMS formation, quorum sensing, increase in population and maturation [29]. Attachment is the initial step for the formation, it starts colonization, and gram + ve bacteria produce EPS. EPS including (DNA, protein, polysaccharides and other inorganic molecule). Quorum sensing is used for cell-to-cell communication [30], and final the maturation step and dispersal. A virulence gene Prf A, a transcriptional activator PrfA, including internalin A and internalin B, internalin A, helps in attachment of bacteria with the surfaces such as stainless steel, glass and polystyrene [31]. Without the expression of internalin A and internalin B, bacteria would not be able to form biofilm. PrfA also help in the formation of EPS, including (DNA, proteins, polysaccharides and other inorganic materials)., formation of EPS is important as it would maintain the bacterial structural integrity and help in the stable biofilm formation. EPS, covers the bacteria against environmental stresses, includes: antimicrobial agents, bactericide agents, germicide agents and oxidative stresses. After attachment and formation of a complete EPA matrix, the transcriptional factor PrfA regulates those stabilizing genes which are used for the formation of the biofilm and would help it to stabilize for a long-time survival. PrfA also regulates some genes that ensure Listeria for the energy production inside biofilm. Sigma B factor which helps the bacteria in the formation of biofilm, sigma B is activated under extremophilic conditions such as; high temperature, high salt, high acid. When conditions become favourable sigma B starts activating and regulating the tolerance gene that would help the bacteria to tolerate harsh conditions. Sigma B produce a gene called bapL (which would use by the bacteria in the initial attachment). Without the attachment by Sigma B Listeria would not able to form biofilm. EPS is important for stable biofilm formation, sigma B provides favourable environment for the growth of Listeria against harsh conditions such as; oxidative stress, heat stress. When the biofilm reached to the maturation stage sigma B help in regulating the genes that responsible for signal production. Signal or cell to cell communication this process is also called Quorum sensing. When biofilm maturation completes, process of dispersal take place, this is important for the new colonization. AgrBDCA, is stands for accessory gene regulation which are Agr B, Agr D, Agr C, Agr A. AgrA and Agr D is used to induce AIP (autoinducing peptides), Agr D would be cleaved by AgrB, for the production of AIP (autoinducing peptides), they act as signalling molecules in Quorum sensing process. Once bacterial cell density reaches to an increase level, agr system become active and AIP binds to Agr C, and promotes biofilm dispersal. AgrC acts as sensor kinase, AgrC binds to AIP and by the process of autophosphorylation AgrC become active. Once it is activated a cascade of reaction parallelly starts, during the reaction one phosphate group of AgrC is transports to AgrA, this reaction is important for regulating the gene that are responsible for the expression of biofilm formation. AgrC is less active in the during the primary stage pf biofilm formation such as (attachment, EMS production, Quorum sensing) but when biofilm reaches at the maturation stage AgrC become active and leads to biofilm dispersal, important for the new colonization. AgrA this is a response regulator by the process of phosphorylation it become activated by AgrC. AgrA is a response regulator [32], regulated those gene that take part in biofilm formation. AgrA help in the formation of EPS, and it can promote the dispersal of biofilm when new colonization takes place, and help in transition from maturation stage to original planktonic stage. Agr system varies with variation in temperature. For example; if the temperature is 25 °C this system is not active and the process of quorum sensing diminished and if the temperature is 37 °C this system is active and AIP level increases which leads to dispersal. If the bacteria form biofilm it can severally affect the host.
Some special features of Listeria include its analysis at the initial level, Listeria has low contamination levels and there is still no specific enumeration method for testing food. It can develop resistance to antibiotics (Gentamycin, Ampicillin) [33,34], resistance to common preservative methods (salt or acidic condition) and cross-resistance to other stresses. It can survive and multiply itself at very low temperature (5 ℃) even can reach dangerous levels during storage. It can grow itself in the presence or absence of oxygen and contaminate food during processing. Many food-born bacteria can survive nearing freezing temperatures, but not able to absorb nutrients, grow and replicate.
Detection of Listeria in food samples
1. Proof man LMTIA method: this is a new novel nucleic acid amplification method for the detection of Listeria in food samples and this technique is based on the principle that the primer for LMTIA were designed based on the target sequence this technique can produce single chain structure, double chain structure and raft chain structure during the amplification stage [35]. The proof man probe is labeled with a fluorophore at 3-prime end and quencher at 5-prime ends respectively a DNA polymerase (pfu) [35] was also acquainted with for the sequence specific detection. It is important to have a mismatch at the 3-prime end of the proof man probe to activate the 3-prime nucleotide and 5-prime exonuclease activity of the proofreading enzyme pfu. After binding of the proof man probe to the target sequences the mismatched nucleotide will be cleaved by the help of the enzyme pfu, it will lead to the release of a fluorophore, slit probe can act as an extensible primer that improves isothermal amplification efficiency [35]. Positive samples in the presence of the target gene can be amplified and detected by a pink fluorescent signal. Negative signals in the absence of the target gene resulted in no amplification and hence no fluorescent signal. Hence on the basis of the proof man probe LMTIA amplification products can be detects by the florescence intensity as the amplification reaction proceeds.
2. Optical label free biosensor: this method does not require labelling such that in (ELISA and PCR in these methods labelling is required for detection and the labelling could be of fluorescence and dye with the target microorganism and consumes lots of time) but this technique is label free thus reduces the preparation time. Label free biosensor [36] is used to detect Listeria in food due to their perceptiveness, preciseness and capabilities to detect Listeria in real time. Optical biosensors identify variations in light properties as they engage with target molecules [36]. In case of L. monocytogenes surface is impregnated with antibodies, aptamers and Molecularly Imprinted Polymers (MIP) that specific for Listeria. In Surface Plasmon Resonance (SPR), when Listeria binds to the surface of biosensors it leads to a change in the refractive index that interchange the resonance angle of the light this swapping is used to detect Listeria in real time, and photonic crystals sensors depends on the interchange in the refractive index when Listeria binds to the surface of the sensors leads an interchange in the wavelength of the reflective light, and interferometric sensors are used to detect the phase interchange of light and it is caused when Listeria binds to the surface of sensors [36]. Optical biosensors enable real time detection of Listeria infection in food without complex sample preparation, this is a very sensitive technique. This technique can detect Listeria even at very low concentration present in any food sample. In milk, meat and vegetables biosensors can directly detect Listeria because these biosensors can be integrated with the microfluidic system to enable a continuous flow of food samples onto the sensor surface and this would lead to high throughput screening of food products.
3. Zero tolerance policy: L. monocytogenes is a pathogenic bacterium that cause serious illness in pregnant women, neonatal and immunocompromised patients. Since ready to eat food products do not undergo cooking more chances of a bacteria to present in the food and as a result the bacteria can easily enters in the humans and cause infection. That is why a zero-tolerance policy is administrated by America., where FDA regulates that no Listeria should be present in 25-gram sample of ready to eat food products and if Listeria is present in these samples that food is considered to be an adulterated and subjected to recall [37].
4. Artificial intelligence: Artificial intelligence contributes a lot in detecting Listeria in foods. Artificial intelligence includes the concept of machine learning and deep learning techniques [38], which are very beneficial to analyze the images by the help of microscopy. AI systems are trained so that it can easily recognize the special characteristic of Listeria, and distinguish it with other food borne pathogens. Under microscopy it significantly speeding up and reduces the rate of human error. AI algorithm helps in identify the colony morphology very accurately than the traditional methods. Hence AI helps enhancing the quality of images [39]. AI algorithms examine the whole genome sequencing data to more effectively detect L. monocytogenes. AI algorithm recognize the specific gene which are responsible for the identification of Listeria [39], and make it feasible to detect and diagnose. AI could process the data from label free biosensors so that it can detect the small changes in the external environment, indication for the presence of Listeria. Some AI integrated sensors and label free biosensor [40], detecting Listeria in the food industries by sensing the electrical signals from the very small changes in the external environment. AI can be used as an early indication weapon. AI system can be integrated to microbial data to detect an early contamination source so that it can used as an early indication weapon.
Treatment and diagnosis of Listeria
Prevention of Listeria could be done by consuming properly cook the food, keeping food at a suitable temperature, washing hands and surfaces before and after cooking the food, remove cheese rinds and pasteurize soft cheese. Listeriosis can be treated by using antibiotics. The most recommended and effective medicine for listeriosis is Ampicillin. It interferes with bacterial cell wall synthesis, shows bactericidal activity. Sometimes, Gentamicin in combination with Ampicillin can be used. Gentamicin is an aminoglycoside antibiotic that interferes with bacterial protein synthesis. Persons who cannot use Ampicillin are recommended to use trimethoprim-sulfamethoxazole. Antibiotics for pregnant women also prevent the foetus from listeriosis. There are two ways to prevent Listeria infections: first, when the bacteria contaminate food, and second, when it infects the body. Antibiotics, such as previously discussed Gentamycin and Ampicillin, are very efficient modern drugs that prevent infections. However, improper and inadequate dosage of antibiotics can lead to bacterial resistance. Bacteriophages, bacteriocins, competitive bacterial species like LAB, plant-based extracts like essential oils, and endolysins are some of the alternative antimicrobial approaches that researchers are currently using at the experimental level to prevent and control the growth of L. monocytogenes in food. While bacteria harm their cell walls, bacteriophages specifically target and lyse bacterial cells. Essential oils specifically target certain enzymes and membrane permeability that prevent bacteria from resisting. However, these alternative methods are only operating at the experimental level, which emphasizes the necessity for more research on the methods being developed rather than relying solely on antibiotics as a main treatment. Antibiotic resistance is an important concern even though antibiotics are the main treatment for Listeria. Resistance may arise from improper antibiotic doses or from taking antibiotics without a prescription. To survive, bacteria can also create biofilms. Bacteria will begin to form biofilms if the dosage is inadequate. Antibiotics cannot properly penetrate these situations, and the bacteria are not eliminated. Bacteria start to get resistant to those antibiotics when they are not eradicated. Additionally, Listeria is an intracellular pathogen, which means it lives within the host cell. Bacteria may potentially acquire resistance if the antibiotic is unable to enter the host cell. Diagnosis is done by taking the following samples for the detection of L. monocytogenes in blood, cerebrospinal fluid [34], vomit, food, animal feed, etc. High throughput techniques are employed for detecting Listeria, it includes pulsed field gel electrophoresis and polymerase chain reaction. These techniques are used to determine DNA fingerprint patterns. These methods directly target nucleic acids and help in delivering more accurate, reliable, and minimizing cases of false positive results. PCR helps to detect the minimum DNA concentration by inoculating meat samples with different inocula of L. monocytogenes.
This review concludes listeriosis is a serious diseased which need to be diagnosed properly by various techniques including whole genome sequencing (gold standard test for Listeria), Enzyme Linked Immunosorbent Assay (ELISA), Polymerase Chain Reaction (PCR), Pulsed- Field Gel Electrophoresis (PFGE). Raw food, raw vegetables and dairy products should not be used without washing. Food must be cooked properly and stored in the refrigerator at low temperature. The species monocytogenes consists of 13 serotypes which includes 1/2a, 1/2b, 1/2c, 3a, 3b, 3c, 4a, 4ab, 4b, 4c, 4d, 4e, and 7. Among 13 serotypes 1/2a, 1/2b, and 4b are related to cause foodborne infections. L. monocytogenes cause foodborne mortality, and 255 deaths recorded in the America and fatality rate 15%-30%. Listeria shows mild symptoms with self-limiting infection and in healthy individuals it remains asymptomatic sometimes. In pregnant females and immunocompromised patients, it identifies by fever, muscle aches, nausea and vomiting with the occasional diarrhoea. It can be a challenging task to recognize the food which caused the infection as the incubation period of the bacteria is too long. Research on this bacterium will provide considerable insight into how pathogenic bacteria adapt to mammalian hosts, invade eukaryotic cells, move intracellularly, interfere with host cell functions and disseminate within tissues.
Declaration
Authors declare there is no competing interest. All the authors have read and approved the MS for its submission to JFST including the concerned authorities.
Authors contribution: Kavita: Writing original draft, making figures and tables; Sakshi Tewari: Writing original draft, conceptualizing, analysis, proof reading and editing.
Highlights
- High throughput methods for detection of Listeria monocytogens in food samples
- National and International outbreaks of L. monocytogens
- Detailed mechanisms of action of L. monocytogens in pregnant females, infants Robustness of L. monocytogens
- Grigore-Gurgu L, Bucur FI, Mihalache OA, Nicolau AI. Comprehensive review on the biocontrol of Listeria monocytogenes in food products. Foods. 2024;13(5):734. Available from: https://doi.org/10.3390/foods13050734
- Bento D, Bomar PA. Listeria monocytogenes. StatPearls Publishing; 2024. Available from: https://pubmed.ncbi.nlm.nih.gov/30521259/
- Farber JM, Peterkin PI. Listeria monocytogenes, a food-borne pathogen. Microbiol Rev. 1991;55(3):476–511. Available from: https://doi.org/10.1128/mr.55.3.476-511.1991
- Osek J, Wieczorek K. Why does Listeria monocytogenes survive in food and food-production environments? J Vet Res. 2023;67(4):537–544. Available from: https://doi.org/10.2478/jvetres-2023-0068
- Desai AN, Anyoha A, Madoff LC, Lassmann B. Changing epidemiology of Listeria monocytogenes outbreaks, sporadic cases, and recalls globally: a review of ProMED reports from 1996 to 2018. Int J Infect Dis. 2019;84:48–53. Available from: https://research.amanote.com/publication/qJ8g3nMBKQvf0BhiLnPG/changing-epidemiology-of-listeria-monocytogenes-outbreaks-sporadic-cases-and-recalls
- Silva A, Silva V, Gomes JP, Coelho A, Batista R, Saraiva C, et al. Listeria monocytogenes from food products and food-associated environments: antimicrobial resistance, genetic clustering and biofilm insights. Antibiotics. 2024;13(5):447. Available from: https://www.oasisbr.ibict.br/vufind/Record/RCAP_7d3d36b1b012da63029deb96d9756923
- Fleming DW, Cochi SL, MacDonald KL, Brondum J, Hayes PS, Plikaytis BD, et al. Pasteurized milk as a vehicle of infection in an outbreak of listeriosis. N Engl J Med. 1985;312:404–407. Available from: https://doi.org/10.1056/nejm198502143120704
- de las Heras A, Cain RJ, Bielecka MK, Vazquez-Boland JA. Regulation of Listeria virulence: PrfA master and commander. Curr Opin Microbiol. 2011;14:118–127. Available from: https://doi.org/10.1016/j.mib.2011.01.005
- Lecuit M. Human listeriosis and animal models. Microbes Infect. 2007;9:1216–1225. Available from: https://doi.org/10.1016/j.micinf.2007.05.009
- Gaillard JL, Berche P, Frehel C, Gouin E, Cossart P. Entry of Listeria monocytogenes into cells is mediated by internalin. Cell. 1991;65:1127–1141. Available from: https://doi.org/10.1016/0092-8674(91)90009-n
- Vazquez-Boland JA, Dominguez-Bernal G, Gonzalez-Zorn B, Kreft J, Goebel W. Pathogenicity islands and virulence evolution in Listeria. Microbes Infect. 2001;3:571–584. Available from: https://doi.org/10.1016/s1286-4579(01)01413-7
- Freitag NE, Port GC, Miner MD. Listeria monocytogenes: from saprophyte to intracellular pathogen. Nat Rev Microbiol. 2009;7(9):623–628. Available from: https://doi.org/10.1038/nrmicro2171
- Robbins JR, Skrzypczynska KM, Zeldovich VB, Kapidzic M, Bakardjiev AI, Schneider DS. Placental syncytiotrophoblast constitutes a major barrier to vertical transmission of Listeria monocytogenes. PLoS Pathog. 2010;6(1):e1000732. Available from: https://doi.org/10.1371/journal.ppat.1000732
- Lecuit M, Nelson DM, Smith SD, Khun H, Huerre M, Vacher-Lavenu MC, et al. Targeting and crossing of the human maternofetal barrier by Listeria monocytogenes. Proc Natl Acad Sci USA. 2004;101(16):6152–6157. Available from: https://research.pasteur.fr/en/publication/targeting-and-crossing-of-the-human-maternofetal-barrier-by-listeria-monocytogenes-role-of-internalin-interaction-with-trophoblast-e-cadherin/
- Vigliani MB, Bakardjiev AI. Intracellular organisms as placental invaders. Fetal Matern Med Rev. 2014;25(3–4):332–338. Available from: https://doi.org/10.1017/S0965539515000066
- Correia de Sá A, Casanova D, Ferreira AL, Fernandes C, Cotter J. Listeriosis in pregnancy: a rare but high-risk infection. Cureus. 2023;15(10):e47748. Available from: https://www.cureus.com/articles/200788-listeriosis-in-pregnancy-a-rare-but-high-risk-infection#!/
- Rowe JH, Ertelt JM, Aguilera MN, Farrar MA, Way SS. Foxp3(+) regulatory T cell expansion required for sustaining pregnancy compromises host defense. Cell Host Microbe. 2011;10(1):54–64. Available from: https://doi.org/10.1016/j.chom.2011.06.005
- Rowe JH, Ertelt JM, Xin L, Way SS. Listeria monocytogenes cytoplasmic entry induces fetal wastage. PLoS Pathog. 2012;8(8):e1002873. Available from: https://doi.org/10.1371/journal.ppat.1002873
- Chaturvedi V, Ertelt JM, Jiang TT, Kinder JM, Xin L, Owens KJ, et al. CXCR3 blockade protects against Listeria monocytogenes-induced fetal wastage. J Clin Invest. 2015;125(4):1713–1725. Available from: https://doi.org/10.1172/jci78578
- Krishnan L, Pejcic-Karapetrovic B, Gurnani K, Zafer A, Sad S. Pregnancy does not deter development of protective CD8+ T-cell responses. Am J Reprod Immunol. 2010;63(1):54–65. Available from: https://doi.org/10.1111/j.1600-0897.2009.00766.x
- Krishnan L, Pejcic-Karapetrovic B, Gurnani K, Zafer A, Sad S. Pregnancy does not deter the development of a potent maternal protective CD8+ T-cell immune response against Listeria monocytogenes. Am J Reprod Immunol. 2010;63(1):54–65. Available from: https://doi.org/10.1111/j.1600-0897.2009.00766.x
- Tachibana M, Hashino M, Nishida T, Shimizu T, Watarai M. Protective role of heme oxygenase-1 in Listeria monocytogenes-induced abortion. PLoS One. 2011;6(9):e25046. Available from: https://doi.org/10.1371/journal.pone.0025046
- Qiu X, Zhu L, Pollard JW. Colony-stimulating factor-1-dependent macrophage functions regulate maternal immune responses against Listeria monocytogenes during early gestation. Infect Immun. 2009;77(1):85–97. Available from: https://doi.org/10.1128/iai.01022-08
- Walker SJ, Archer P, Banks JG. Growth of Listeria monocytogenes at refrigeration temperatures. J Appl Bacteriol. 1990;68:157–162. Available from: https://doi.org/10.1111/j.1365-2672.1990.tb02561.x
- Bucur FI, Grigore-Gurgu L, Crauwels P, Riedel CU, Nicolau AI. Resistance of Listeria monocytogenes to stress conditions encountered in food and food processing environments. Front Microbiol. 2018;9:2700. Available from: https://doi.org/10.3389/fmicb.2018.02700
- Cotter PD, Hill C. Surviving the acid test: responses of gram-positive bacteria to low pH. Microbiol Mol Biol Rev. 2003;67(3):429–453. Available from: https://doi.org/10.1128/mmbr.67.3.429-453.2003
- Soares CA, Knuckley B. Mechanistic studies of the agmatine deiminase from Listeria monocytogenes. Biochem J. 2016;473:1553–1561. Available from: https://doi.org/10.1042/bcj20160221
- Byun KH, Kim HJ. Survival strategies of Listeria monocytogenes under environmental stress: biofilm formation and stress responses. Food Sci Biotechnol. 2023;32(12):1631–1651. Available from: https://doi.org/10.1007/s10068-023-01427-6
- Yang Y, Kong X, Niu B, Yang J, Chen Q. Differences in biofilm formation of Listeria monocytogenes and their effects on virulence and drug resistance. Foods. 2024;13(7):1076. Available from: https://doi.org/10.3390/foods13071076
- West KHJ, Ma SV, Pensinger DA, Tucholski T, Tiambeng TN, Eisenbraun EL, et al. Characterization of an autoinducing peptide signal in quorum sensing and biofilm formation in Listeria monocytogenes. Biochemistry. 2023;62(19):2878–2892. Available from: https://doi.org/10.1021/acs.biochem.3c00373
- Finn L, Onyeaka H, O’Neill S. Listeria monocytogenes biofilms in food-associated environments: a persistent enigma. Foods. 2023;12(18):3339. Available from: https://doi.org/10.3390/foods12183339
- Miller MB, Bassler BL. Quorum sensing in bacteria. Annu Rev Microbiol. 2001;55:165–199. Available from: https://doi.org/10.1146/annurev.micro.55.1.165
- Baquero F, Lanza VF, Duval M, Coque TM. Ecogenetics of antibiotic resistance in Listeria monocytogenes. Mol Microbiol. 2020;113(3):570–579. Available from: https://doi.org/10.1111/mmi.14454
- Bäckman A, Lantz P, Rådström P, Olcén P. Evaluation of an extended diagnostic PCR assay for detection of bacterial meningitis. Mol Cell Probes. 1999;13:49–60. Available from: https://doi.org/10.1006/mcpr.1998.0218
- Song C, Wang B, Wang Y, Liu J, Wang D. Detection of Listeria monocytogenes using the Proofman-LMTIA assay. Molecules. 2023;28(14):5457. Available from: https://doi.org/10.3390/molecules28145457
- Fernández-Blanco A, Hernández-Pérez M, Moreno-Trigos Y, García-Hernández J. Development of optical label-free biosensor method for detection of Listeria monocytogenes. Sensors (Basel). 2023;23(12):5570. Available from: https://doi.org/10.3390/s23125570
- Churchill RL, Lee H, Hall JC. Detection of Listeria monocytogenes and listeriolysin O in food. J Microbiol Methods. 2006;64(2):141–170. Available from: https://doi.org/10.1016/j.mimet.2005.10.007
- Ma L, Yi J, Wisuthiphaet N, Earles M, Nitin N. Accelerating detection of bacteria in food using artificial intelligence and optical imaging. Appl Environ Microbiol. 2023;89(1):e0182822. Available from: https://aifs.ucdavis.edu/news/accelerating-the-detection-of-bacteria-in-food-using-ai-and-optical-imaging
- Hameed S, Xie L, Ying Y. Conventional and emerging detection techniques for pathogenic bacteria in food science: a review. Trends Food Sci Technol. 2018;81:61–73. Available from: https://www.sciencedirect.com/science/article/abs/pii/S0924224417307689
- Tanaka T, Kogiso A, Maeda Y, Matsunaga T. Colony fingerprinting: a novel method for discrimination of food-contaminating microorganisms based on bioimage informatics. In: Proc IEEE Int Symp Circuits Syst. 2019. Available from: https://ieeexplore.ieee.org/document/8702644