More Information
Submitted: March 10, 2026 | Accepted: March 23, 2026 | Published: March 24, 2026
Citation: Nasta P, Di Gregorio D, Zaia M, Carosi G. Gut Microbiota Alterations Associated with Viral Activity in Chronic Hepatitis B. Arch Case Rep. 2026; 10(3): 21-29. Available from:
https://dx.doi.org/10.29328/journal.acr.1001185
DOI: 10.29328/journal.acr.1001185
Copyright license: © 2026 Nasta P, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Keywords: Chronic Hepatitis B (CHB); Gut microbiota dysbiosis; Viral activity; Gut–liver axis; Microbial diversity; Pathobionts; Short-Chain Fatty Acids (SCFAs); Microbiota-targeted therapy
Gut Microbiota Alterations Associated with Viral Activity in Chronic Hepatitis B
Paola Nasta1,2*, Marco Di Gregorio3, Monica Zaia1 and Giampiero Carosi2
1FdS Association (The Thread of Health E.T.S. Third Sector Organization), Italy
2MISI Foundation (Infectious Diseases and International Health), Italy
3University of Brescia Spedali, Civili General Hospital, Italy
*Corresponding author: Paola Nasta, FdS Association (The Theard of Health E.T.S. Third Sector Entity), MISI Foundation (Infectious Diseases and International Health), Italy, Email: [email protected]
Background: The gut–liver axis plays a pivotal role in chronic liver diseases, and increasing evidence links gut microbiota dysbiosis to disease progression in chronic hepatitis B (CHB). However, data comparing microbiota features between patients with active and inactive HBV infection remain limited.
Methods: We performed a retrospective analysis of gut microbiota composition and functional potential in patients with chronic HBV infection, stratified according to viral activity as defined by current EASL guidelines. Microbial diversity, taxonomic composition, enterotypes, pathobiont prevalence, and predicted metabolic pathways were compared between active and inactive HBV infection.
Results: Patients with active HBV infection exhibited significantly reduced microbial α-diversity, lower phylogenetic diversity, and decreased evenness compared with inactive carriers, despite virological suppression under antiviral therapy. Active HBV was associated with Prevotella-dominated enterotypes, expansion of pathobionts (Escherichia coli, Klebsiella pneumoniae, Bacteroides fragilis), and depletion of beneficial symbionts, particularly short-chain fatty acid– and indole-producing taxa, including Faecalibacterium prausnitzii, Roseburia intestinalis, Blautia wexlerae, and Alistipes spp. Functional profiling revealed reduced production of butyrate, propionate, acetate, and indole derivatives, alongside increased synthesis of ethanol, secondary bile acids, and lipopolysaccharides.
Conclusion: Active CHB is characterized by a distinct dysbiotic and pro-inflammatory gut microbiota profile that persists despite effective antiviral therapy. These findings support a contributory role of gut microbiota in immune dysregulation and disease activity in CHB and provide a rationale for microbiota-targeted interventions as complementary therapeutic strategies.
Growing evidence supports the existence of a close and bidirectional relationship between the gut and the liver, commonly referred to as the gut–liver axis, which integrates metabolic, immunological, and inflammatory pathways. The gut microbiota plays a central role in this axis, exerting metabolic, immune, neurological, and nutritional functions, and interacting continuously with the liver. Alterations in gut microbial composition (dysbiosis) have been increasingly associated with liver injury and disease progression [1-3].
In recent years, significant advances have been made in elucidating the relationship between chronic hepatitis B (CHB) and the gut microbiota. An increasing number of studies have demonstrated that gut microbiota dysbiosis is associated with disease progression in patients with CHB [4-7]. Moreover, emerging evidence suggests that therapeutic strategies targeting dysbiosis may represent a promising adjunct in the management of chronic liver diseases, including HBV infection [1-3].
Microbiota alterations appear to be particularly pronounced in advanced stages of HBV-related liver disease. In patients with hepatocellular carcinoma (HCC), a distinct gut microbiota signature has been described, with 122 taxa differing significantly from healthy controls. Among these, Bacteroides, Veillonella, Phenylobacterium, Synechococcus, and u114 were the most enriched genera. At the phylum level, an increased Bacteroidetes/Firmicutes ratio and a higher abundance of Proteobacteria were observed in patients with cirrhosis and HCC. Furthermore, Haemophilus, Fusobacterium, Veillonella, Streptococcus, and Ruminococcus increased progressively with disease severity [4,5].
A reduction in gut microbial alpha diversity has consistently been associated with disease progression, with the lowest diversity reported in cirrhotic patients. Specific bacterial species, such as Dialister succinatiphilus and Alistipes onderdonkii, are abundant in healthy individuals but markedly reduced across successive stages of CHB [6]. Additionally, Bifidobacterium abundance is significantly reduced in patients with CHB and further decreased in cirrhosis, whereas members of the Enterobacteriaceae family are enriched in advanced disease stages [7].
Data regarding the relationship between gut microbiota composition and the ability to control HBV viremia remain limited. Experimental models, however, suggest a pivotal role of the microbiota in antiviral immune responses. In murine models, the absence of gut microbiota or Toll-like receptor 4 (TLR4) signaling impairs Kupffer cell activation, reduces interleukin-10 (IL-10) production, and compromises viral clearance [8,9]. Antibiotic-induced depletion of gut bacteria in adult mice results in impaired HBV clearance, associated with reduced CD4⁺ follicular helper T-cell–mediated support of germinal center B-cell differentiation [10].
Short-chain fatty acids (SCFAs), including acetate, propionate, and butyrate, are key microbial metabolites that sustain intestinal epithelial integrity and immune homeostasis [31].
Dysbiosis-associated reductions in butyrate-producing taxa—such as Alistipes, Bacteroides, Ruminococcus, and Anaerostipes—have been linked to increased intestinal permeability and hepatic inflammation in CHB. Notably, Anaerostipes abundance has been reported to be higher in inactive HBV carriers with normal alanine aminotransferase (ALT) levels compared with patients with active disease, supporting a link between gut microbial composition and hepatic inflammatory activity [11].
Increased intestinal permeability has been documented in patients with CHB. Serum levels of zonulin, a regulator of tight junction integrity, and copeptin have been shown to correlate inversely with HBV DNA levels. Elevated zonulin levels have been observed in HBV-infected patients with HCC compared with those with CHB alone, cirrhosis, or healthy controls [12,13]. Disruption of the intestinal barrier facilitates bacterial translocation and systemic inflammation. In patients with CHB and decompensated cirrhosis, increased concentrations of secretory immunoglobulin A (sIgA) and tumor necrosis factor (TNF)-α have been reported, with Enterobacteriaceae abundance correlating positively with sIgA levels, while Bifidobacterium shows a weak negative correlation with TNF-α [14].
Bile acids represent another critical link between the gut and the liver. While most bile acids are reabsorbed in the enterohepatic circulation, a fraction is metabolized by the gut microbiota into secondary bile acids, which exert metabolic and immunomodulatory effects. Alterations in bile acid profiles have been associated with liver fibrosis progression, with impaired conversion of primary to secondary bile acids observed in patients with more advanced fibrosis stages [15].
Translocation of oral microbiota to the gut has also been described in cirrhotic patients, including those with HBV-related disease. Taxa such as Eikenella corrodens, Gemella haemolysans, Leptotrichia, Fusobacterium periodonticum, Selenomonas spp., Actinomyces odontolyticus, and Streptococcus moorei may further contribute to intestinal and hepatic inflammation [16].
Lipopolysaccharide (LPS), a component of the cell wall of Gram-negative bacteria, is a potent driver of systemic and hepatic inflammation. Increased intestinal permeability promotes LPS translocation into the portal circulation, activating pro-inflammatory pathways in the liver and contributing to fibrosis progression. Elevated LPS levels have been associated with disease severity in HBV-related liver failure [17,18]. The relative increase in Gram-negative bacteria, particularly Bacteroides, along with a reduction in Gram-positive Firmicutes, may underlie the increased circulating LPS burden observed in CHB.
To date, however, data comparing gut microbiota characteristics between patients with active versus inactive hepatitis B remain scarce. Therefore, we conducted a retrospective analysis to characterize gut microbiota features in HBV-infected individuals and to identify microbiota-related differences associated with viral activity, as defined by current EASL guidelines [19].
Study setting and population
FdS Il Filo della Salute ETS is a third-sector, non-profit organization dedicated to integrative medicine and involved in training, information, and health education activities. Gut microbiota analysis is offered to individuals who access the services provided by the association.
Sociodemographic and clinical data are routinely collected in a dedicated database and retrospectively analyzed after written informed consent is obtained. From the entire database, individuals affected by hepatitis B (HBsAg-positive) were identified, and a retrospective observational study was planned.
Inclusion criteria were: age >18 years; HBsAg positivity; availability of gut microbiota analysis performed after informed consent; active follow-up at a referral clinical center; absence of significant liver fibrosis (F0–F1), with liver stiffness values between 5 and 7 kPa.
Sociodemographic, clinical, therapeutic, and microbiota-related data of subjects meeting inclusion and exclusion criteria and stored in the FdS database were included in the analysis.
Gut microbiota analysis
Gut micorbiota analysis was performed using the Wellmicro® test (Wellmicro, Italy), a commercial metagenomic platform based on next-generation sequencing (NGS) of microbial DNA extracted from stool samples.
Stool samples were self-collected using a standardized collection kit provided by the manufacturer, which allows stabilization of microbial DNA at room temperature for up to several weeks before processing.
According to the manufacturer, the analysis is based on high-throughput sequencing approaches (including shotgun metagenomics in updated versions of the test) followed by proprietary bioinformatic pipelines for taxonomic classification and functional inference.
The output includes relative abundance of microbial taxa, biodiversity indices (alpha and beta diversity), and composite indices such as the Firmicutes/Bacteroidetes ratio and dysbiosis index.
Due to the proprietary nature of the platform, detailed information regarding DNA extraction protocols, sequencing parameters, and bioinformatic workflows is not fully disclosed by the provider.
Study objectives
The primary objective of the study was to describe gut microbiota characteristics in HBsAg-positive patients.
The secondary objective was to compare gut microbiota features between HBsAg-positive individuals with active hepatitis B (active HBV) receiving antiviral therapy and those with inactive hepatitis B (inactive HBV) not receiving antiviral treatment. The definition of active and inactive hepatitis B was based on the most recent EASL guidelines [19].
All patients were followed at dedicated hepatology referral centers.
The following analyses were performed:
- Descriptive analysis of baseline characteristics (sociodemographic variables, risk factors, and clinical features) of HBsAg-positive patients stratified into active HBV and inactive HBV groups.
- Description and comparison of gut microbiota characteristics between active HBV and inactive HBV populations, including: dysbiosis index; biodiversity index; biodiversity parameters (number of species, phylogenetic diversity—PD whole tree, species distribution, Pielou’s evenness index); enterotype; Firmicutes/Bacteroides ratio; Prevotella/Bacteroides ratio; bacterial ecology (relative abundance of the most represented phyla); and presence of pathobionts.
- Evaluation of the number of subjects showing a normal representation of butyrate-producing symbionts, including Faecalibacterium prausnitzii, Roseburia intestinalis, Eubacterium ramulus ATCC 29099, Blautia wexlerae, and Alistipes spp.
- Description of the metabolic potential of the active HBV versus inactive HBV populations, including analysis of acetate, butyrate, propionate, indoleacetic acid (IAA), indolepropionic acid (IPA), tryptamine, lipopolysaccharide (LPS), secondary bile acids, and ethanol.
Statistical analysis
Descriptive analysis was performed using medians and standard deviations for continuous variables and absolute numbers and percentages for categorical variables. A retrospective descriptive statistical analysis was conducted to compare populations, with particular attention to baseline sociodemographic characteristics.
For each variable, absolute and relative frequencies were calculated for categorical data, while continuous variables were reported as mean and standard deviation or median and interquartile range, according to data distribution. Differences between groups were assessed using the chi-square test (or Fisher’s exact test, when appropriate) for categorical variables and the independent-samples t-test or Mann–Whitney U test for continuous variables, depending on data normality as assessed by the Shapiro–Wilk test.
Study population
The overall study sample included 50 subjects, divided into two groups: 25 individuals with active hepatitis B (active HBV) and 25 individuals with inactive hepatitis B (inactive HBV). Baseline characteristics of the study population are summarized in Table 1.
| Table1: Baselinecharacteristics of the enrolled sample. | ||||
| TotalHBv+ (N 50) | HBV-aCTIVE(N 25) | HBV-INACTIVE(N 25) | RR(95%IC) p | |
| Sex (%) | ||||
| Female | 30(60) | 16(64) | 14(56) | 1,1(0.6-2,1) |
| Male | 20(40) | 9(36) | 11(44) | (0.8(0.4-1.5) |
| Age Year (median IQR) | 72(55-80) | 71(58-80) | 69(55-75) | 0.6 |
| Feeding (n%) | ||||
| maternal | 30(60) | 10(40) | 20(80) | 0.4(0.2-0.7)0.008 |
| Formula | 17(34) | 14(56) | 3(12) | 2.4(1.4-4.1)0.002 |
| Both | 3(6) | 1(4) | 2(8) | 1.3(0.5-3.1) |
| Alcohol use | ||||
| Daily n% | 5(10) | 3(12) | 2(8) | 0.9(0.7-1.3) |
| Montly (n%) | 10(20) | 5(20) | 5(20) | 1.2(0.5-2.6) |
| Weekly (n%) | 3(6) | 2(8) | 1(4) | 1.3(0.5-3.1.) |
| NO alcoohl | 22(44) | 15(60) | 17(68) | 0.8(0.4-1.4) |
| Birth | ||||
| Naturalbirth (n%) | 31(62) | 13(52) | 18(72) | 0.4(0.1-1.3) |
| Diet before visit (n%) | ||||
| Lactose free | 11(22) | 5(20) | 6(24) | 0.8(0.4-1.8) |
| Symptoms at MycRobiota test execution (n%) | ||||
| Anxiety | ||||
| Depression | 28(56) | 18(72) | 10(40) | 2.0(1.3-2.9)0.04 |
| Memory disorders | 17(34) | 12(48) | 5(20) | 1.7(1.0-3.0)0.03 |
| Sleep disorders | 4(8) | 22(88) | 19(76) | 1.6(0.6-4.2) |
| Dermatitis | 31(62) | 17(68) | 14(56) | 1.3(0.7-1.4) |
| Abdominalswelling/pain | 5(10) | 3(12) | 2(8) | 1.2(0.5-2.6) |
| Constipation | 33(66) | 21(84) | 12(48) | 2.7(1.1-6.6)0.01 |
| Alternating bowelhabits | 21(42) | 12(48) | 9(36) | 1.2(0.7-1.2) |
| Urinary infections | 29(58) | 19(76) | 10(40) | 2.2(1.1-4.7)0.02 |
| Fatigue | 15(30) | 9(36) | 6(24) | 1.3(0.7-2.2) |
| 31(62) | 19(76) | 11(44) | 2.2(1.1-4.3)0.04 | |
| Comorbidities (n%) | ||||
| Type 2 diabetes | 26(52) | 17(68) | 9(36) | 1.9(1.0-3.6 |
| NASH | 40(80) | 22(88) | 18(72) | 1.8(0.6-4.9) |
| Cardiovascular dis. | 22(44) | 15(60) | 7(28) | 1.9(1.0-3.3)0.04 |
| Diverticulosis | 23(46) | 17(68) | 6(24) | 1.4(2.3-4.6)0.04 |
| Arthralgia | 28(56) | 21(84) | 7(28) | 4.1(1.6-10.2)0.0001 |
| Allergies | 5(10) | 2(8) | 3(12) | 0.7(0.2-2.3) |
| Thyroid disease | 9(18) | 14(56) | 5(20) | 0.4(0.1-0.9)0.01 |
| Fibrosi (n%) | ||||
| F0(<7kps) | 5(10) | 2(8) | 3(12) | 0.7(0.2-2.3) |
| F 1 | 27(54) | 12(48) | 15(60) | 0.7(0.4-1.3) |
| F2 | 10(20) | 6(24) | 4(16) | 1.2(0.6-2.3) |
| F3-4 | 8(16) | 5(20) | 3(12) | 1.6(0.7-3.3) |
| Drugs assumption (n%) | ||||
| PPI | 45(90) | 23(92) | 24(96) | 0.7(0.3-1.7) |
| FANS | 11(22) | 5(20) | 6(24) | 0.8(0.4-1.8) |
| Entecavir | 12(24) | 12(48) | 0 | |
| tenofovir | 13(26) | 13(52) | 0 | |
The median age of the cohort was 72 years (IQR 55–80) and was comparable between the two groups (71 vs. 69 years; p = 0.6). Overall, 60% of the participants were female (64% in the active HBV group and 56% in the inactive HBV group; p = 0.7).
A history of breastfeeding was more frequently reported among subjects with inactive HBV compared with those with active HBV (80% vs. 40%; RR 0.4, 95% CI 0.2–0.7; p = 0.008). No significant differences were observed in the type of delivery, which was vaginal in 52% of subjects with active HBV and 72% of those with inactive HBV (RR 0.4, 95% CI 0.1–1.3).
Alcohol consumption was similar in the two groups; most subjects reported abstinence from alcohol, in accordance with hepatoctlogist recommendations for patients with chronic liver disease. A lactose-free diet was followed by a minority of subjects (20% in the active HBV group and 25% in the inactive HBV group; RR 0.8, 95% CI 0.4–1.8).
Self-reported symptoms at the time of gut microbiota analysis differed significantly between groups. Anxiety was reported by 72% of subjects with active HBV compared with 40% of those with inactive HBV (RR 2.0, 95% CI 1.3–2.9; p = 0.04). Depression was reported by 48% of subjects with active HBV and 20% of those with inactive HBV (RR 1.7, 95% CI 1.0–3.0; p = 0.03).
Abdominal pain and bloating were reported by 84% of subjects with active HBV and 48% of those with inactive HBV (RR 2.7, 95% CI 1.1–6.6; p = 0.01). Alternating bowel habits were reported by 36% of active HBV subjects compared with 24% of inactive HBV subjects (RR 2.2, 95% CI 1.1–4.7; p = 0.02). Fatigue was significantly more frequent in subjects with active HBV than in those with inactive HBV (76% vs. 44%; RR 2.2, 95% CI 1.1–4.3; p = 0.04).
Cardiovascular diseases (RR 1.9, 95% CI 1.0–3.3; p = 0.04), diverticulosis (RR 1.4, 95% CI 2.3–4.6; p = 0.04), arthralgia (RR 4.1, 95% CI 1.6–10.2; p = 0.0001), and thyroid disorders (RR 0.4, 95% CI 0.1–0.9; p = 0.01) were more frequently reported among subjects with active HBV compared with those with inactive HBV.
No significant differences were observed in the use of proton pump inhibitors, gastroprotective agents, or non-steroidal anti-inflammatory drugs. Liver fibrosis stage, assessed by the most recent available FibroScan measurement reported by participants, did not differ between active and inactive HBV groups.
All subjects with active HBV were receiving antiviral therapy: 48% were treated with entecavir and 52% with tenofovir.
Gut microbiota characteristics
In the analyzed cohort, the number of observed species—a direct indicator of taxonomic richness—was lower in subjects with active HBV compared with those with inactive HBV (median 81 [IQR 63–89] vs. 118 [IQR 90–110] species), although this difference did not reach statistical significance (p = 0.1).
Median phylogenetic diversity (PD whole tree) was significantly lower in the active HBV group compared with the inactive HBV group (12.7 [IQR 10.6–16.3] vs. 20.4 [IQR 13.9–21.3]; p = 0.001). Similarly, analysis of Pielou’s evenness index showed significantly lower values in subjects with active HBV compared with those with inactive HBV (0.3 [IQR 0.6–0.8] vs. 0.7 [IQR 0.6–0.9]; p = 0.03).
The Firmicutes/Bacteroides ratio (physiological reference range 0.3–3.2) did not differ significantly between groups. Median values were comparable (active HBV: 1.4 [IQR 0.9–2.3] vs. inactive HBV: 1.3 [IQR 0.8–2.1]; p = 0.6). In contrast, the Prevotella/Bacteroides ratio was significantly higher in subjects with active HBV (1.02 [IQR 0.8–2.19]) compared with those with inactive HBV (0.1 [IQR 0.0–0.4]; p = 0.02) (Table 2).
| Table 2:biodiversityindices and distribution of microbial species. | |||
| Variabiles | HBV Active(n25) | HBVInactive(n25) | RR(95%IC)p |
| Observedspecies number (median IQR) | 81(63–89) | 118(90–110) | 0.1 |
| Phylogeneticdistance(median IQR) | 12.7 (10.6–16.3) | 20.4(13.9–21.3) | 0.001 |
| Species distribution (Pielou's index, median, IQR) | 0.3(0.6–0.8) | 0.7(0.6–0.9) | 0.03 |
| Firmicutes/Bacteroides ratio (median IQR) | 1,4(0.9-2.3) | 1.3(0.8-2.1) | 0.6 |
| Prevotella/Bacteroides(median IQR) | 1.02(0.8-2.3) | 0.1(0.0-0.4) | 0.02 |
| Enterotypes | |||
| EnterotipoBcateroides (n%) | 9(36) | 8(32) | 1.0(0.6-1.9) |
| Enterotipo Prevotella (n%) | 12(48) | 5(20) | 1.7(1.0-3.0)0.07 |
| Enterotipo Faecalibacterium(n%) | 2(3) | 7(28) | 0.3(0.1-2.3) |
| Other | 2(3) | 5(20) | 0.5(0.1-21.7) |
| Patientswith identified pathobionts(n%) | 14(56) | 7(28) | 1.7(1.0-3.0)0.02 |
| Patientswith normal range of Butyrate producing bacteria | |||
| Faecalibacterium prausnitzii (n%) | 8(32) | 19(76) | 0.4(0.2-0.7)0.004 |
| Roseburia intestinalis | 7(28) | 18(72) | 0.3(0.1-0.7)0.004 |
| Eubacterium ramulus ATCC 29099 | 8(32) | 16(64) | 0.5(0.2-0.9)0.04 |
| Blautia wexlerae | 9(36) | 15(60) | 0.6(0.3-1.1)0.05 |
| Alistipes species | 6(24) | 17(68) | 0.3(0.1-0.7)0.004 |
| Bacteroides (%) | 38.6 (25.5–44.9) | 33.6(25.8–45.4) | 0.8 |
| Firmicutes (%) | 51.2(43.1–59.7) | 49.6(37.6–59.5) | 0.6 |
| Actinobacteria (%) | 2.9 (1.6–5.6) | 3.3(2.0–4.6) | 0.6 |
| Verrucomicrobia (%) | 0.0(0.0–0.6) | 0.0(0.0–1.1) | 0.2 |
| Proteobacteria (%) | 3.8 (1.5–5.3) | 2.6(1.0–4.1) | 0.3 |
Phylum-level composition and enterotypes
Enterotype analysis revealed heterogeneity across groups. The Bacteroides-dominated enterotype was more frequent in subjects with inactive HBV (9 subjects, 36%) than in those with active HBV (8 subjects, 32%), although this difference was not statistically significant (RR 1.0, 95% CI 0.6–1.9). Conversely, the Prevotella enterotype was significantly more frequent in subjects with active HBV (12 subjects, 44%) compared with those with inactive HBV (5 subjects, 20%) (RR 1.7, 95% CI 1.01–3.0; p = 0.07). The Faecalibacterium enterotype and the “other” category, including mixed or unclassifiable enterotypes, did not differ significantly between groups.
Pathobionts (potentially opportunistic or pathogenic bacteria) were identified in 14 subjects (56%) with active HBV and in 7 subjects (28%) with inactive HBV (RR 1.7, 95% CI 1.0–3.0; p = 0.02). In the active HBV group, Bacteroides fragilis (6 patients), Escherichia coli (5 patients), and Klebsiella pneumoniae (4 patients) were identified. In the inactive HBV group, Bacteroides fragilis (3 patients), Candida albicans (2 patients), and Escherichia coli (2 patients) were detected.
At the phylum level, differences in relative abundance did not reach statistical significance. Mean relative abundance of Bacteroidetes was slightly higher in subjects with active HBV (38.6%, IQR 25.5–44.9) compared with inactive HBV subjects (33.6%, IQR 25.8–45.4; p = 0.8). Firmicutes abundance was comparable between groups (active HBV: 51.2%, IQR 43.1–59.7 vs. inactive HBV: 49.6%, IQR 37.6–59.5; p = 0.6). Actinobacteria showed slightly lower median values in the active HBV group (2.9%, IQR 1.6–5.6) compared with the inactive HBV group (3.3%, IQR 2.0–4.6; p = 0.6). Verrucomicrobia and Proteobacteria, although present at low relative abundances, did not differ significantly between groups (p = 0.2 and p = 0.3, respectively).
Analysis of butyrate-producing bacterial families
Butyrate-producing bacteria are considered key biomarkers of intestinal health. Among the most relevant taxa is Faecalibacterium prausnitzii (family Ruminococcaceae, phylum Firmicutes, class Clostridia; physiological relative abundance range in the applied test: 0.26–7.3%). The proportion of subjects with values within the normal range was significantly lower in the active HBV group compared with the inactive HBV group (32% vs. 76%; RR 0.4, 95% CI 0.2–0.7; p = 0.004).
Similar findings were observed for Roseburia intestinalis (physiological range 0.06–2.9%), Eubacterium ramulus ATCC 29099 (0.01–0.56%), and Blautia wexlerae (0.8–8.41%), all belonging to the Lachnospiraceae family (phylum Firmicutes, class Clostridia). These taxa were present within the normal range in a significantly lower proportion of subjects with active HBV compared with inactive HBV subjects: 28% vs. 72% (RR 0.3, 95% CI 0.1–0.7; p = 0.004), 32% vs. 64% (RR 0.5, 95% CI 0.2–0.9; p = 0.04), and 36% vs. 60% (RR 0.6, 95% CI 0.3–1.1; p = 0.05), respectively.
Several species of the genus Alistipes (family Rikenellaceae, phylum Bacteroidetes, class Bacteroidia) were identified in the study population. However, a significantly higher proportion of subjects with inactive HBV showed values within the normal range compared with subjects with active HBV (68% vs. 24%; RR 0.3, 95% CI 0.1–0.7; p = 0.004).
Metabolic potential analysis
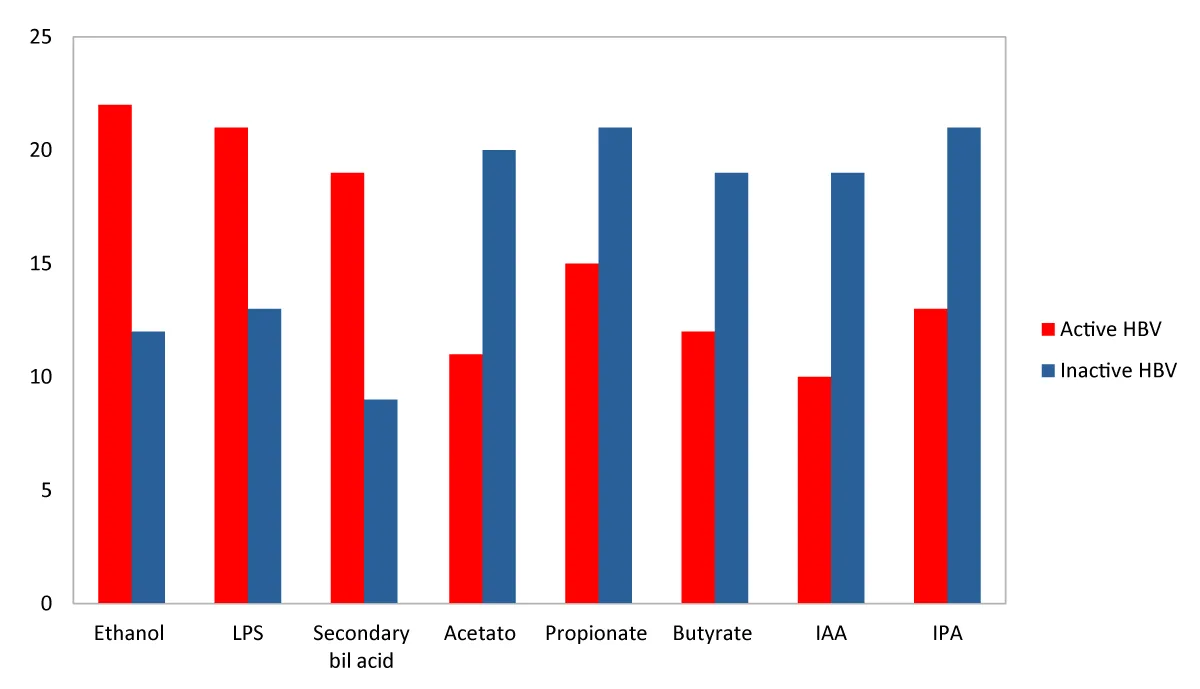
Compared with subjects with inactive HBV, those with active HBV showed a reduced production of butyrate (48% vs. 76%; RR 0.2, 95% CI 0.08–0.9; p = 0.005), propionate (60% vs. 84%; RR 0.5, 95% CI 0.3–0.9; p = 0.03), and acetate (80% vs. 44%; RR 0.1, 95% CI 0.5–0.6; p = 0.01).
Conversely, a higher proportion of subjects with active HBV exhibited excess production of secondary bile acids (76% vs. 36%; RR 2.4, 95% CI 1.2–5.1; p = 0.009), ethanol (88% vs. 48%; RR 3.4, 95% CI 1.2–9.8; p = 0.005), and lipopolysaccharides (84% vs. 52%; RR 2.4, 95% CI 1.0–6.0; p = 0.03).
Finally, 10 (40%) and 13 (52%) subjects with active HBV, compared with 19 (76%) and 21 (84%) subjects with inactive HBV, showed insufficient production of indoleacetic acid (IAA) (RR 0.4, 95% CI 0.2–0.8; p = 0.02) and indolepropionic acid (IPA) (RR 0.5, 95% CI 0.3–0.8; p = 0.03), respectively (Figure 1).
Figure 1: Differences in gut microbiota metabolic activity between subjects with active and inactive HBV infection. Subjects with active HBV infection showed reduced production of short-chain fatty acids, including butyrate, propionate, and acetate, compared with those with inactive HBV. Conversely, increased production of secondary bile acids, ethanol, and lipopolysaccharides was observed in active HBV. In addition, subjects with active HBV more frequently exhibited insufficient production of indole derivatives, including indoleacetic acid (IAA) and indolepropionic acid (IPA).
The role of the gut microbiota in chronic hepatitis B (CHB) has become increasingly relevant, not only for understanding disease pathogenesis but also for elucidating mechanisms involved in viral control and fibrosis progression. In the present study, we compared gut microbiota composition and functional potential in patients with active and inactive HBV infection, identifying distinct microbial and metabolic profiles associated with viral activity.
Patients with active HBV infection displayed a significant reduction in microbial diversity compared with inactive carriers, despite sustained virological suppression under antiviral therapy. Similar findings have been reported in recent multi-omics studies, suggesting that gut dysbiosis persists independently of serum HBV-DNA levels and reflects a chronic inflammatory milieu associated with HBV-related liver disease [20,21]. The absence of differences in dietary habits between groups supports the hypothesis that host–virus–microbiota interactions, rather than lifestyle factors, drive these alterations.
Phylogenetic diversity, assessed using the PD whole tree index, was significantly lower in patients with active HBV. This index captures the evolutionary and functional breadth of the microbial ecosystem, indicating that although species richness may be partially preserved, the microbiota in active HBV is composed of phylogenetically closer taxa with overlapping metabolic functions. Such a configuration reflects a functionally impoverished microbiota with reduced resilience and adaptive capacity, which may contribute to the persistence of low-grade systemic inflammation, as previously described in HBV-associated dysbiosis [20].
Consistently, Pielou’s evenness index was reduced in active HBV patients, suggesting an uneven microbial structure dominated by selected taxa. Reduced evenness has been associated with impaired immune tolerance and increased susceptibility to inflammatory perturbations in chronic liver diseases [21].
While the Firmicutes/Bacteroidetes ratio remained within physiological ranges in both groups, the Prevotella/Bacteroides ratio was higher in patients with active HBV. Prevotella-enriched microbiota profiles have been associated with pro-inflammatory immune responses and enhanced mucosal immune activation. This shift was further supported by enterotype analysis, showing a higher prevalence of Prevotella-dominated enterotypes among active HBV patients.
A significantly higher proportion of subjects with active HBV harbored pathobionts, including Escherichia coli, Klebsiella pneumoniae, and Bacteroides fragilis. Expansion of opportunistic taxa has been linked to impaired gut barrier function and increased immune activation through gut-associated lymphoid tissue, favoring disease progression [21]. These findings are consistent with previous observations of increased Enterobacteriaceae abundance in advanced liver disease and hepatocellular carcinoma (HCC) [22-26].
Despite comparable phylum-level composition, qualitative alterations in beneficial symbionts distinguished active from inactive HBV infection. In particular, active HBV patients showed a marked reduction in butyrate-producing bacteria, including Faecalibacterium prausnitzii, Roseburia intestinalis, Eubacterium ramulus, and Blautia wexlerae. These taxa play a central role in maintaining intestinal epithelial integrity, regulating immune tolerance, and suppressing inflammation through short-chain fatty acid (SCFA) production.
Notably, species belonging to the genus Alistipes were significantly reduced in active HBV patients. Alistipes spp., anaerobic commensals commonly detected in healthy individuals, have been progressively linked to liver fibrosis severity across different liver diseases [22-26]. Their reduction has also been observed in NAFLD and NASH, suggesting a common fibrogenic microbiota signature [27]. Alistipes spp. contribute to acetate and propionate production, and their depletion may impair SCFA availability, promoting intestinal permeability and hepatic inflammation [28].
Experimental evidence supports this hypothesis, as restoration of Alistipes abundance through probiotic supplementation or fiber-rich diets has been associated with increased SCFA production, improved gut barrier integrity, and reduced hepatic Th17 recruitment via decreased LPS translocation [29-31].
Functional metabolic analysis further reinforced the taxonomic findings. Patients with active HBV exhibited reduced production of butyrate, propionate, and acetate, alongside increased production of ethanol, secondary bile acids, and lipopolysaccharides (LPS). Excess endogenous ethanol production by gut bacteria has been recently described in HBV-associated dysbiosis and may act as a cofactor accelerating liver injury [32]. Similarly, altered bile acid metabolism reflects impaired microbial transformation capacity and has been associated with fibrosis progression and inflammatory signaling.
Additionally, active HBV patients showed significantly reduced production of the tryptophan-derived microbial metabolites indole-3-acetic acid (IAA) and indole-3-propionic acid (IPA). These indoles exert protective effects on intestinal barrier integrity and modulate host immune responses by suppressing NF-κB signaling and endotoxin-induced inflammation. Reduced availability of IAA and IPA has been associated with worsened steatosis and inflammation in metabolic liver disease models [33], supporting their role as key mediators along the gut–liver axis.
Overall, our findings define a distinct dysbiotic profile in active HBV infection characterized by reduced phylogenetic diversity, depletion of immunoregulatory symbionts, expansion of pathobionts, and a metabolic shift toward pro-inflammatory and hepatotoxic products. These alterations persist despite antiviral-induced viral suppression, suggesting that gut microbiota may contribute to disease pathophysiology rather than representing a secondary epiphenomenon.
This study demonstrates that active chronic hepatitis B is associated with a profound qualitative and functional alteration of the gut microbiota, independent of lifestyle factors and despite effective virological suppression. Dysbiosis in active HBV is characterized by reduced microbial diversity, loss of beneficial SCFA- and indole-producing symbionts, increased intestinal permeability, and expansion of pathobionts with enhanced endotoxin production.
This condition is not merely a consequence of liver disease but could also contribute to immune dysregulation. An altered gut microbiota compromises regulatory T-cell and dendritic cell homeostasis, potentially impairing immune-mediated control of HBV replication [34,35]. Reduced microbial diversity and disruption of tight junction integrity facilitate bacterial translocation and portal influx of pathogen-associated molecular patterns, particularly LPS [34,35].
The continuous exposure of the liver to bacterial products overstimulates Kupffer cells and hepatic immune infiltrates through Toll-like receptor activation, amplifying non-specific inflammation while paradoxically failing to achieve viral clearance. This immune “anarchy” promotes hepatocellular injury, necroinflammation, and fibrosis progression [35,36].
Fibrosis progression in HBV infection should therefore be interpreted as a multifactorial process driven by the synergistic interaction between viral persistence and microbiota-derived inflammatory signals. Bacterial translocation has been proposed as a “second hit,” exacerbating tissue damage mediated by an immune system unable to distinguish between antiviral defense and systemic inflammation [37-39].
Taken together, these findings support the concept that modulation of the gut microbiota may represent a complementary therapeutic strategy in chronic hepatitis B, aimed at restoring immune balance, reinforcing intestinal barrier integrity, and limiting inflammation-driven liver injury. Longitudinal and interventional studies are warranted to clarify whether microbiota-targeted approaches can improve clinical outcomes in HBV infection [40].
Given the observational nature of this study, these findings should be interpreted as associations, and the proposed mechanisms remain speculative [41].
We would like to thank Gilead Sciences for their unconditional contribution.
In memory of Professor Giampiero Caorsi, whose contribution to this work and to our scientific growth will not be forgotten.
Funding statement
This study received a publication grant from Gilead Sciences, awarded to Il Filo della Salute ETS, which supported the publication costs.
The funding body had no role in the study design, data collection, analysis, interpretation, or manuscript preparation.
Conflict of interest statement: The authors declare no competing interests. Funding support from Gilead Sciences was provided solely for publication purposes.
- Kang Y, Cai Y. Gut microbiota and hepatitis-B-virus-induced chronic liver disease: implications for faecal microbiota transplantation therapy. J Hosp Infect. 2017;96(4):342-348. Available from: https://doi.org/10.1016/j.jhin.2017.04.007
- Tranah TH, Edwards LA, Schnabl B, Shawcross DL. Targeting the gut-liver-immune axis to treat cirrhosis. Gut. 2020;70(5):982-994. Available from: https://doi.org/10.1136/gutjnl-2020-320786
- Fukui H. Gut microbiome-based therapeutics in liver cirrhosis: basic consideration for the next step. J Clin Transl Hepatol. 2017;5(3):249-260. Available from: https://doi.org/10.14218/jcth.2017.00008
- Yang J, He Q, Lu F, Chen K, Ni Z, Wang H, et al. A distinct microbiota signature precedes the clinical diagnosis of hepatocellular carcinoma. Gut Microbes. 2023;15(1):2201159. Available from: https://doi.org/10.1080/19490976.2023.2201159
- Zeng Y, Chen S, Fu Y, Wu W, Chen T, Chen J, et al. Gut microbiota dysbiosis in patients with hepatitis B virus-induced chronic liver disease, covering chronic hepatitis, liver cirrhosis, and hepatocellular carcinoma. J Viral Hepat. 2020;27(2):143-155. Available from: https://doi.org/10.1111/jvh.13216
- Chen Z, Xie Y, Zhou F, Zhang B, Wu J, Yang L, et al. Featured gut microbiomes associated with the progression of chronic hepatitis B disease. Front Microbiol. 2020;11:383. Available from: https://doi.org/10.3389/fmicb.2020.00383
- Lu H, Wu Z, Xu W, Yang J, Chen Y, Li L. Intestinal microbiota was assessed in cirrhotic patients with hepatitis B virus infection. Microb Ecol. 2011;61(3):693-703. Available from: https://doi.org/10.1007/s00248-010-9801-8
- Chou HH, Chien WH, Wu LL, Cheng CH, Chung CH, Horng JH, et al. Age-related immune clearance of hepatitis B virus infection requires the establishment of gut microbiota. Proc Natl Acad Sci U S A. 2015;112(7):2175-2180. Available from: https://doi.org/10.1073/pnas.1424775112
- Xu L, Yin W, Sun R, Wei H, Tian Z. Kupffer cell-derived IL-10 plays a key role in maintaining humoral immune tolerance in hepatitis B virus-persistent mice. Hepatology. 2014;59(2):443-452. Available from: https://doi.org/10.1002/hep.26668
- Wu T, Li F, Chen Y, Wei H, Tian Z, Sun C, et al. CD4(+) T cells play a critical role in microbiota-maintained anti-HBV immunity in a mouse model. Front Immunol. 2019;10:927. Available from: https://doi.org/10.3389/fimmu.2019.00927
- Wang G, Huang S, Wang Y, Cai S, Yu H, Liu H, et al. Bridging intestinal immunity and gut microbiota by metabolites. Cell Mol Life Sci. 2019;76(20):3917-3937. Available from: https://doi.org/10.1007/s00018-019-03190-6
- Wang X, Li MM, Niu Y, Zhang X, Yin JB, Zhao CJ, et al. Serum zonulin in HBV-associated chronic hepatitis, liver cirrhosis, and hepatocellular carcinoma. Dis Markers. 2019;2019:5945721. Available from: https://doi.org/10.1155/2019/5945721
- Calgin MK, Cetinkol Y. Decreased levels of serum zonulin and copeptin in chronic hepatitis B patients. Pak J Med Sci. 2019;35(3):847-851. Available from: https://doi.org/10.12669/pjms.35.3.144
- Liu Q, Li F, Zhuang Y, Xu J, Wang J, Mao X, et al. Alteration in gut microbiota associated with hepatitis B and non-hepatitis virus related hepatocellular carcinoma. Gut Pathog. 2019;11:1. Available from: https://doi.org/10.1186/s13099-018-0281-6
- Caussy C, Hsu C, Singh S, Bassirian S, Kolar J, Faulkner C, et al. Serum bile acid patterns are associated with the presence of NAFLD in twins, and dose-dependent changes with increase in fibrosis stage in patients with biopsy-proven NAFLD. Aliment Pharmacol Ther. 2019;49(2):183-193. Available from: https://agris.fao.org/search/en/providers/122439/records/6798e03ad6a63682f045f442
- Chen Y, Ji F, Guo J, Shi D, Fang D, Li L. Dysbiosis of small intestinal microbiota in liver cirrhosis and its association with etiology. Sci Rep. 2016;6:34055. Available from: https://doi.org/10.1038/srep34055
- Pan C, Gu Y, Zhang W, Zheng Y, Peng L, Deng H, et al. Dynamic changes of lipopolysaccharide levels in different phases of acute-on-chronic hepatitis B liver failure. PLoS One. 2012;7(11):e49460. Available from: https://doi.org/10.1371/journal.pone.0049460
- Deng YD, Peng XB, Zhao RR, Ma CQ, Li JN, Yao LQ. The intestinal microbial community dissimilarity in hepatitis B virus-related liver cirrhosis patients with and without alcohol consumption. Gut Pathog. 2019;11:58. Available from: https://doi.org/10.1186/s13099-019-0337-2
- European Association for the Study of the Liver. EASL Clinical Practice Guidelines on the management of hepatitis B virus infection. J Hepatol. 2025;83(2):502-583. Available from: https://doi.org/10.1016/j.jhep.2025.03.018
- Shen Y, Wu SD, Chen Y, Li XY, Zhu Q, Nakayama K, et al. Alterations in gut microbiome and metabolomics in chronic hepatitis B infection-associated liver disease and their impact on peripheral immune response. Gut Microbes. 2023;15(1):2155018. Available from: https://doi.org/10.1080/19490976.2022.2155018
- Yan F, Zhang Q, Shi K, Zhang Y, Zhu B, Bi Y, et al. Gut microbiota dysbiosis with hepatitis B virus liver disease and association with immune response. Front Cell Infect Microbiol. 2023;13:1152987. Available from: https://doi.org/10.3389/fcimb.2023.1152987
- Rautio M, Eerola E, Väisänen-Tunkelrott ML, Molitoris D, Lawson P, Collins MD, et al. Reclassification of Bacteroides putredinis in a new genus Alistipes gen. nov., as Alistipes putredinis comb. nov., and description of Alistipes finegoldii sp. nov., from human sources. Syst Appl Microbiol. 2003;26(2):182-188. Available from: https://doi.org/10.1078/072320203322346029
- Shkoporov AN, Chaplin AV, Khokhlova EV, Shcherbakova VA, Motuzova OV, Bozhenko VK, et al. Alistipes inops sp. nov. and Coprobacter secundus sp. nov., isolated from human feces. Int J Syst Evol Microbiol. 2015;65:4580-4588. Available from: https://doi.org/10.1099/ijsem.0.000617
- Iebba V, Guerrieri F, Di Gregorio V, Levrero M, Gagliardi A, Santangelo F, et al. Combining amplicon sequencing and metabolomics in cirrhotic patients highlights distinctive microbiota features involved in bacterial translocation, systemic inflammation and hepatic encephalopathy. Sci Rep. 2018;8:8210. Available from: https://doi.org/10.1038/s41598-018-26509-y
- Shao L, Ling Z, Chen D, Liu Y, Yang F, Li L. Disorganized gut microbiome contributed to liver cirrhosis progression: a meta-omics-based study. Front Microbiol. 2018;9:3166. Available from: https://doi.org/10.3389/fmicb.2018.03166
- Sung CM, Lin YF, Chen KF, Ke HM, Huang HY, Gong YN, et al. Predicting clinical outcomes of cirrhosis patients with hepatic encephalopathy from the fecal microbiome. Cell Mol Gastroenterol Hepatol. 2019;8:301-318.e2. Available from: https://doi.org/10.1016/j.jcmgh.2019.04.008
- Rau M, Rehman A, Dittrich M, Groen AK, Hermanns HM, Seyfried F, et al. Fecal SCFAs and SCFA-producing bacteria in gut microbiome of human NAFLD as a putative link to systemic T-cell activation and advanced disease. United Eur Gastroenterol J. 2018;6:1496-1507. Available from: https://doi.org/10.1177/2050640618804444
- Oliphant K, Allen-Vercoe E. Macronutrient metabolism by the human gut microbiome: major fermentation by-products and their impact on host health. Microbiome. 2019;7:91. Available from: https://doi.org/10.1186/s40168-019-0704-8
- Li J, Sung CY, Lee N, Ni Y, Pihlajamäki J, Panagiotou G, et al. Probiotics modulate gut microbiota suppresses hepatocellular carcinoma growth in mice. Proc Natl Acad Sci U S A. 2016;113:E1306-E1315. Available from: https://doi.org/10.1073/pnas.1518189113
- Bindels LB, Porporato P, Dewulf EM, Verrax J, Neyrinck AM, Martin JC, et al. Gut microbiota-derived propionate reduces cancer cell proliferation in the liver. Br J Cancer. 2012;107:1337-1344. Available from: https://doi.org/10.1038/bjc.2012.409
- Koh A, De Vadder F, Kovatcheva-Datchary P, Bäckhed F. From dietary fiber to host physiology: short-chain fatty acids as key bacterial metabolites. Cell. 2016;165(6):1332-1345. Available from: https://doi.org/10.1016/j.cell.2016.05.041
- Magdy Wasfy R, Mbaye B, Borentain P, Tidjani Alou M, Murillo Ruiz ML, Caputo A, Andrieu C, Armstrong N, Million M, Gerolami R. Ethanol-producing Enterocloster bolteae is enriched in chronic hepatitis B-associated gut dysbiosis: a case-control culturomics study. Microorganisms. 2023;11(10):2437. Available from: https://doi.org/10.3390/microorganisms11102437
- Min BH, Devi S, Kwon GH, Gupta H, Jeong JJ, Sharma SP, et al. Gut microbiota-derived indole compounds attenuate metabolic dysfunction-associated steatotic liver disease by improving fat metabolism and inflammation. Gut Microbes. 2024;16(1):2307568. Available from: https://doi.org/10.1080/19490976.2024.2307568
- Chou YC, Chen CL, Sheen IS. The gut ecosystem and the pathogenesis of chronic hepatitis B virus infection. Clin Microbiol Infect. 2015;21(6):548-553.
- Yan F, Zhang Q, Shi K, Zhang Y, Zhu B, Bi Y, Wang X. Gut microbiota dysbiosis with hepatitis B virus liver disease and association with immune response. Front Cell Infect Microbiol. 2023;13:1152987. Available from: https://doi.org/10.3389/fcimb.2023.1152987
- Seki E, Schnabl B. Role of innate immunity and the microbiota in liver fibrosis: crosstalk between the liver and gut. J Physiol. 2012;590(3):447-458. Available from: https://doi.org/10.1113/jphysiol.2011.219691
- Wiest R, Albillos A, Trauner M, Bajaj JS, Jalan R. Targeting the gut-liver axis in liver disease. J Hepatol. 2017;67(5):1084-1103. Available from: https://doi.org/10.1016/j.jhep.2017.05.007
- Dapito DH, Mencin A, Gwak GY, Pradere JP, Jang MK, Mederacke I, et al. Promotion of hepatocellular carcinoma by the intestinal microbiota and TLR4. Cancer Cell. 2012;21(4):504-516. Available from: https://doi.org/10.1016/j.ccr.2012.02.007
- Parada Venegas D, De la Fuente MK, Landskron G, González MJ, Quera R, Dijkstra G, Harmsen HJM, et al. Short-chain fatty acids (SCFAs)-mediated gut epithelial and immune regulation and its relevance for inflammatory bowel diseases. Front Immunol. 2019;10:277. Available from: https://doi.org/10.3389/fimmu.2019.00277
- Tripathi A, Debelius J, Brenner DA, Karin M, Loomba R, Schnabl B, Knight R. The gut-liver axis and the intersection with the microbiome. Nat Rev Gastroenterol Hepatol. 2018;15(7):397-411. Available from: https://doi.org/10.1038/s41575-018-0011-z
- Lynch SV, Pedersen O. The human intestinal microbiome in health and disease. N Engl J Med. 2016;375(24):2369-2379. Available from: https://doi.org/10.1056/nejmra1600266